Active calcium accumulation underlies severe weakness in a panel of mice with slow-channel syndrome
- PMID: 12151524
- PMCID: PMC6758155
- DOI: 10.1523/JNEUROSCI.22-15-06447.2002
Active calcium accumulation underlies severe weakness in a panel of mice with slow-channel syndrome
Abstract
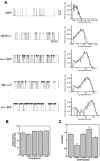
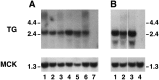
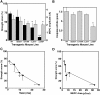
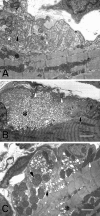
Mutations affecting the gating and channel properties of ionotropic neurotransmitter receptors in some hereditary epilepsies, in familial hyperekplexia, and the slow-channel congenital myasthenic syndrome (SCCMS) may perturb the kinetics of synaptic currents, leading to significant clinical consequences. Although at least 12 acetylcholine receptor (AChR) mutations have been identified in the SCCMS, the altered channel properties critical for disease pathogenesis in the SCCMS have not been identified. To approach this question, we investigated the effect of different AChR subunit mutations on muscle weakness and the function and viability of neuromuscular synapses in transgenic mice. Targeted expression of distinct mutant AChR subunits in skeletal muscle prolonged the decay phases of the miniature endplate currents (MEPCs) over a broad range. In addition, both muscle strength and the amplitude of MEPCs were lower in transgenic lines with greater MEPC duration. SCCMS is associated with calcium overload of the neuromuscular junctional sarcoplasm. We found that the extent of calcium overload of motor endplates in the panel of transgenic mice was influenced by the relative permeability of the mutant AChRs to calcium, on the duration of MEPCs, and on neuromuscular activity. Finally, severe degenerative changes at the motor endplate (endplate myopathy) were apparent by electron microscopy in transgenic lines that displayed the greatest activity-dependent calcium overload. These studies demonstrate the importance of control of the kinetics of AChR channel gating for the function and viability of the neuromuscular junction.
Figures


References
-
- Bhattacharyya BJ, Day J, Gundeck JE, Leonard S, Wollmann RW, Gomez C. Desensitization of mutant acetylcholine receptors in transgenic mice reduces the amplitude of synaptic currents. Synapse. 1997;27:367–377. - PubMed
-
- Bodensteiner JB, Engel AG. Intracellular calcium accumulation in Duchenne dystrophy and other myopathies: a study of 567,000 muscle fibers in 114 biopsies. Neurology. 1978;28:439–446. - PubMed
-
- Brenner HR, Witzemann V, Sakmann B. Imprinting of acetylcholine receptor messenger RNA accumulation in mammalian neuromuscular synapses. Nature. 1990;344:544–547. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
