Differential composition of 5-hydroxytryptamine3 receptors synthesized in the rat CNS and peripheral nervous system
- PMID: 12151552
- PMCID: PMC6758137
- DOI: 10.1523/JNEUROSCI.22-15-06732.2002
Differential composition of 5-hydroxytryptamine3 receptors synthesized in the rat CNS and peripheral nervous system
Abstract
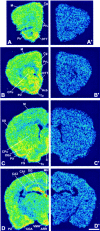
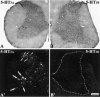
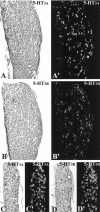
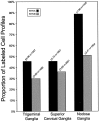
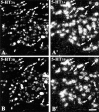
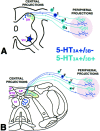
The type 3 serotonin (5-HT3) receptor is the only ligand-gated ion channel receptor for serotonin in vertebrates. Two 5-HT3 receptor subunits have been cloned, subunit A (5-HT3A) and subunit B (5-HT3B). We used in situ hybridization histochemistry and reverse transcriptase-PCR amplification to demonstrate that 5-HT3A subunit transcripts are expressed in central and peripheral neurons. In contrast, 5-HT3B subunit transcripts are restricted to peripheral neurons. Thus, the prevalent form of 5-HT3 receptor synthesized within the CNS lacks the 5-HT3B subunit. Because coexpression of 5-HT3A and 5-HT3B subunits produces heteromeric 5-HT3A/3B receptors with properties that differ from those of 5-HT3A homomeric receptors, we investigated possible coexpression of both subunits at the cellular level. We found that near to 90% of all 5-HT3B expressing neurons coexpress the 5-HT3A subunit in superior cervical and nodose ganglia (NG). In addition, there is a cellular population that expresses only the 5-HT3A subunit. Therefore, peripheral neurons have the capacity to synthesize two different 5-HT3 receptors, 5-HT3A+/3B- and 5-HT3A+/3B+ receptors. We also determined that neurons of NG projecting to the nucleus tractus solitarium and those of dorsal root ganglia projecting to superficial layers of the spinal cord express 5-HT3A or 5-HT3A/3B subunits. Thus, presynaptic 5-HT3 receptors containing the 5-HT3B subunit might be present in these target brain areas. The compartmentalized structural composition of the 5-HT3 receptor may be the basis of functional diversity within this receptor. This raises the possibility that 5-HT3 receptors participating in sympathetic, parasympathetic and sensory functions may be functionally different from those involved in cognition and emotional behavior.
Figures



Similar articles
-
5-HT(3)-receptor subunits A and B are co-expressed in neurons of the dorsal root ganglion.J Comp Neurol. 2001 Sep 17;438(2):163-72. doi: 10.1002/cne.1307. J Comp Neurol. 2001. PMID: 11536186
-
The 5-HT3B subunit is a major determinant of serotonin-receptor function.Nature. 1999 Jan 28;397(6717):359-63. doi: 10.1038/16941. Nature. 1999. PMID: 9950429
-
Cannabinoid CB1 receptor and serotonin 3 receptor subunit A (5-HT3A) are co-expressed in GABA neurons in the rat telencephalon.J Comp Neurol. 2004 Jan 6;468(2):205-16. doi: 10.1002/cne.10968. J Comp Neurol. 2004. PMID: 14648680
-
The 5-hydroxytryptamine type 3 (5-HT3) receptor reveals a novel determinant of single-channel conductance.Biochem Soc Trans. 2004 Jun;32(Pt3):547-52. doi: 10.1042/BST0320547. Biochem Soc Trans. 2004. PMID: 15157181 Review.
-
The 5-HT3 receptor--the relationship between structure and function.Neuropharmacology. 2009 Jan;56(1):273-84. doi: 10.1016/j.neuropharm.2008.08.003. Epub 2008 Aug 12. Neuropharmacology. 2009. PMID: 18761359 Free PMC article. Review.
Cited by
-
Peripheral neural targets in obesity.Br J Pharmacol. 2012 Jul;166(5):1537-58. doi: 10.1111/j.1476-5381.2012.01951.x. Br J Pharmacol. 2012. PMID: 22432806 Free PMC article. Review.
-
Agonist- and antagonist-induced up-regulation of surface 5-HT3 A receptors.Br J Pharmacol. 2015 Aug;172(16):4066-77. doi: 10.1111/bph.13197. Epub 2015 Jul 6. Br J Pharmacol. 2015. PMID: 25989383 Free PMC article.
-
Prolongation of bronchopulmonary C-fiber-mediated apnea by prenatal nicotinic exposure in rat pups: role of 5-HT3 receptors.FASEB J. 2019 Oct;33(10):10731-10741. doi: 10.1096/fj.201900279RR. Epub 2019 Jun 27. FASEB J. 2019. PMID: 31251077 Free PMC article.
-
5-HT3 Receptors in Rat Dorsal Root Ganglion Neurons: Ca2+ Entry and Modulation of Neurotransmitter Release.Life (Basel). 2022 Aug 2;12(8):1178. doi: 10.3390/life12081178. Life (Basel). 2022. PMID: 36013357 Free PMC article.
-
Novel Striatal GABAergic Interneuron Populations Labeled in the 5HT3a(EGFP) Mouse.Cereb Cortex. 2016 Jan;26(1):96-105. doi: 10.1093/cercor/bhu179. Epub 2014 Aug 21. Cereb Cortex. 2016. PMID: 25146369 Free PMC article.
References
-
- Andrews PL, Rapeport WG, Sanger GJ. Neuropharmacology of emesis induced by anti-cancer therapy. Trends Pharmacol Sci. 1988;9:334–341. - PubMed
-
- Barnes JM, Barnes NM, Costall B, Ironside JW, Naylor RJ. Identification and characterization of 5-hydroxytryptamine3 recognition sites in human brain tissue. J Neurochem. 1989;53:1787–1793. - PubMed
-
- Belelli D, Balcarek JM, Hope AG, Peters JA, Lambert JJ, Blackburn TP. Cloning and functional expression of a human 5-hydroxytryptamine type 3As receptor subunit. Mol Pharmacol. 1995;48:1054–1062. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
