Locus-specific control of asymmetric and CpNpG methylation by the DRM and CMT3 methyltransferase genes
- PMID: 12151602
- PMCID: PMC139913
- DOI: 10.1073/pnas.162371599
Locus-specific control of asymmetric and CpNpG methylation by the DRM and CMT3 methyltransferase genes
Abstract
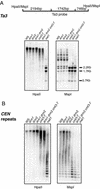
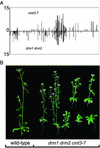
Many plant, animal, and fungal genomes contain cytosine DNA methylation in asymmetric sequence contexts (CpHpH, H = A, T, C). Although the enzymes responsible for this methylation are unknown, it has been assumed that asymmetric methylation is maintained by the persistent activity of de novo methyltransferases (enzymes capable of methylating previously unmodified DNA). We recently reported that the DOMAINS REARRANGED METHYLASE (DRM) genes are required for de novo DNA methylation in Arabidopsis thaliana because drm1 drm2 double mutants lack the de novo methylation normally associated with transgene silencing. In this study, we have used bisulfite sequencing and Southern blot analysis to examine the role of the DRM loci in the maintenance of asymmetric methylation. At some loci, drm1 drm2 double mutants eliminated all asymmetric methylation. However, at the SUPERMAN locus, asymmetric methylation was only completely abolished in drm1 drm2 chromomethylase 3 (cmt3) triple mutant plants. drm1 drm2 double mutants also showed a strong reduction of CpNpG (n = A, T, C, or G) methylation at some loci, but not at others. The drm1 drm2 cmt3 triple mutant plants did not affect CpG methylation at any locus tested, suggesting that the primary CpG methylases are encoded by the MET1 class of genes. Although neither the drm1 drm2 double mutants nor the cmt3 single mutants show morphological defects, drm1 drm2 cmt3 triple mutant plants show pleiotropic effects on plant development. Our results suggest that the DRM and CMT3 genes act in a partially redundant and locus-specific manner to control asymmetric and CpNpG methylation.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

