Measurement of cytoplasmic calcium concentration in the rods of wild-type and transducin knock-out mice
- PMID: 12154183
- PMCID: PMC2290451
- DOI: 10.1113/jphysiol.2001.013987
Measurement of cytoplasmic calcium concentration in the rods of wild-type and transducin knock-out mice
Abstract
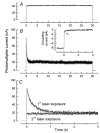
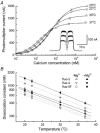
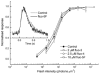
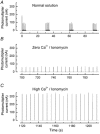
A 10 microm spot of argon laser light was focused onto the outer segments of intact mouse rods loaded with fluo-3, fluo-4 or fluo-5F, to estimate dark, resting free Ca(2+) concentration ([Ca(2+)](i)) and changes in [Ca(2+)](i) upon illumination. Dye concentration was adjusted to preserve the normal physiology of the rod, and the laser intensity was selected to minimise bleaching of the fluorescent dye. Wild-type mouse rods illuminated continuously with laser light showed a progressive decrease in fluorescence well fitted by two exponentials with mean time constants of 154 and 540 ms. Rods from transducin alpha-subunit knock-out (Tralpha-/-) animals showed no light-dependent decline in fluorescence but exhibited an initial rapid component of fluorescence increase which could be fitted with a single exponential (tau~1-4 ms). This fluorescence increase was triggered by rhodopsin bleaching, since its amplitude was reduced by pre-exposure to bright bleaching light and its time constant decreased with increasing laser intensity. The rapid component was however unaffected by incorporation of the calcium chelator BAPTA and seemed therefore not to reflect an actual increase in [Ca(2+)](i). A similar rapid increase in fluorescence was also seen in the rods of wild-type mice just preceding the fall in fluorescence produced by the light-dependent decrease in [Ca(2+)](i). Dissociation constants were measured in vitro for fluo-3, fluo-4 and fluo-5F with and without 1 mM Mg(2+) from 20 to 37 degrees C. All three dyes showed a strong temperature dependence, with the dissociation constant changing by a factor of 3-4 over this range. Values at 37 degrees C were used to estimate absolute levels of rod [Ca(2+)](i). All three dyes gave similar values for [Ca(2+)](i) in wild-type rods of 250 +/- 20 nM in darkness and 23 +/- 2 nM after exposure to saturating light. There was no significant difference in dark [Ca(2+)](i) between wild-type and Tralpha-/- animals.
Figures

Similar articles
-
Time course and magnitude of the calcium release induced by bright light in salamander rods.J Physiol. 2002 Aug 1;542(Pt 3):829-41. doi: 10.1113/jphysiol.2001.013218. J Physiol. 2002. PMID: 12154182 Free PMC article.
-
Early receptor current of wild-type and transducin knockout mice: photosensitivity and light-induced Ca2+ release.J Physiol. 2004 Jun 15;557(Pt 3):821-8. doi: 10.1113/jphysiol.2004.064014. Epub 2004 Apr 8. J Physiol. 2004. PMID: 15073279 Free PMC article.
-
A light-dependent increase in free Ca2+ concentration in the salamander rod outer segment.J Physiol. 2001 Apr 15;532(Pt 2):305-21. doi: 10.1111/j.1469-7793.2001.0305f.x. J Physiol. 2001. PMID: 11306652 Free PMC article.
-
Bleached pigment produces a maintained decrease in outer segment Ca2+ in salamander rods.J Gen Physiol. 1998 Jan;111(1):53-64. doi: 10.1085/jgp.111.1.53. J Gen Physiol. 1998. PMID: 9417134 Free PMC article.
-
Light-dependent changes in outer segment free-Ca2+ concentration in salamander cone photoreceptors.J Gen Physiol. 1999 Feb;113(2):267-77. doi: 10.1085/jgp.113.2.267. J Gen Physiol. 1999. PMID: 9925824 Free PMC article.
Cited by
-
Depleted Calcium Stores and Increased Calcium Entry in Rod Photoreceptors of the Cacna2d4 Mouse Model of Cone-Rod Dystrophy RCD4.Int J Mol Sci. 2022 Oct 28;23(21):13080. doi: 10.3390/ijms232113080. Int J Mol Sci. 2022. PMID: 36361866 Free PMC article.
-
Elevated Ca2+ sparklet activity during acute hyperglycemia and diabetes in cerebral arterial smooth muscle cells.Am J Physiol Cell Physiol. 2010 Feb;298(2):C211-20. doi: 10.1152/ajpcell.00267.2009. Epub 2009 Oct 21. Am J Physiol Cell Physiol. 2010. PMID: 19846755 Free PMC article.
-
A G86R mutation in the calcium-sensor protein GCAP1 alters regulation of retinal guanylyl cyclase and causes dominant cone-rod degeneration.J Biol Chem. 2019 Mar 8;294(10):3476-3488. doi: 10.1074/jbc.RA118.006180. Epub 2019 Jan 8. J Biol Chem. 2019. PMID: 30622141 Free PMC article.
-
A novel Ca2+-feedback mechanism extends the operating range of mammalian rods to brighter light.J Gen Physiol. 2015 Oct;146(4):307-21. doi: 10.1085/jgp.201511412. J Gen Physiol. 2015. PMID: 26415569 Free PMC article.
-
Photopigment quenching is Ca2+ dependent and controls response duration in salamander L-cone photoreceptors.J Gen Physiol. 2010 Apr;135(4):355-66. doi: 10.1085/jgp.200910394. Epub 2010 Mar 15. J Gen Physiol. 2010. PMID: 20231373 Free PMC article.
References
-
- Baylor SM, Hollingworth S. Measurement and interpretation of cytoplasmic [Ca2+] signals from calcium-indicator dyes. News in Physiological Sciences. 2000;15:19–26. - PubMed
-
- Calvert PD, Govardovskii VI, Krasnoperova N, Anderson RE, Lem J, Makino CL. Membrane protein diffusion sets the speed of rod phototransduction. Nature. 2001;411:90–94. - PubMed
-
- Calvert PD, Krasnoperova NV, Lyubarsky AL, Isayama T, Nicolo M, Kosaras B, Wong G, Gannon KS, Margolskee RF, Sidman RL, Pugh EN, Jr, Makino CL, Lem J. Phototransduction in transgenic mice after targeted deletion of the rod transducin alpha-subunit. Proceedings of the National Academy of Sciences of the USA. 2000;97:13913–13918. - PMC - PubMed
-
- Chen CK, Burns ME, He W, Wensel TG, Baylor DA, Simon MI. Slowed recovery of rod photoresponse in mice lacking the GTPase accelerating protein RGS9–1. Nature. 2000;403:557–560. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

