Excitability changes in human peripheral nerve axons in a paradigm mimicking paired-pulse transcranial magnetic stimulation
- PMID: 12154192
- PMCID: PMC2290455
- DOI: 10.1113/jphysiol.2002.018937
Excitability changes in human peripheral nerve axons in a paradigm mimicking paired-pulse transcranial magnetic stimulation
Abstract
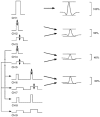
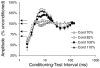
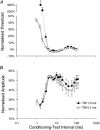
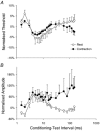
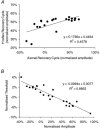
A peripheral nerve model was developed to determine whether changes in axonal excitability could affect the findings in studies of cortical processes using paired-pulse transcranial magnetic stimulation (TMS). The recovery of axonal excitability from a conditioning stimulus smaller than the test stimulus was qualitatively similar to that with suprathreshold conditioning stimuli. There was an initial decrease in excitability, equivalent to refractoriness at conditioning-test intervals < 4 ms, an increase in excitability, equivalent to supernormality, at intervals of 5-20 ms and a second phase of decreased excitability, equivalent to late subnormality at intervals > 30 ms. H reflex studies using conditioning stimuli below threshold for the H reflex established that these excitability changes could be faithfully translated across an excitatory synapse. Changing membrane potential by injecting polarising current altered axonal excitability in a predictable way, and produced results similar to those reported for many disease states using paired-pulse TMS. Specifically, axonal hyperpolarisation produced a smaller decrease in excitability followed by a greater increase in excitability. This study supports the view that changes in excitability of the stimulated axons should be considered before synaptic mechanisms are invoked in the interpretation of findings from paired-pulse TMS studies.
Figures

References
-
- Awiszus F, Feistner H, Urbach D, Bostock H. Characterisation of paired-pulse transcranial magnetic stimulation conditions yielding intracortical inhibition or I-wave facilitation using a threshold-hunting paradigm. Experimental Brain Research. 1999;129:317–324. - PubMed
-
- Bostock H, Baker M. Evidence for two types of potassium channel in human motor axons in vivo. Brain Research. 1988;462:354–358. - PubMed
-
- Bostock H, Cikurel K, Burke D. Threshold tracking techniques in the study of human peripheral nerve. Muscle and Nerve. 1998;21:137–158. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

