Ensheathing cells and methylprednisolone promote axonal regeneration and functional recovery in the lesioned adult rat spinal cord
- PMID: 12177207
- PMCID: PMC6757894
- DOI: 10.1523/JNEUROSCI.22-16-07111.2002
Ensheathing cells and methylprednisolone promote axonal regeneration and functional recovery in the lesioned adult rat spinal cord
Abstract
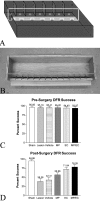
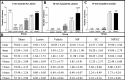
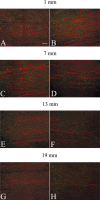
Axons fail to regenerate after spinal cord injury (SCI) in adult mammals, leading to permanent loss of function. After SCI, ensheathing cells (ECs) promote recovery in animal models, whereas methylprednisolone (MP) promotes neurological recovery in humans. In this study, the effectiveness of combining ECs and MP after SCI was investigated for the first time. After lesioning the corticospinal tract in adult rats, ECs were transplanted into the lesion, and MP was administered for 24 hr. At 6 weeks after injury, functional recovery was assessed by measuring successful performance of directed forepaw reaching (DFR), expressed as percentages. Axonal regeneration was analyzed by counting the number of corticospinal axons, anterogradely labeled with biotin dextran tetramethylrhodamine, caudal to the lesion. Lesioned control rats, receiving either no treatment or vehicle, had abortive axonal regrowth (1 mm) and poor DFR success (38 and 42%, respectively). Compared with controls, MP-treated rats had significantly more axons 7 mm caudal to the lesion, and DFR performance was significantly improved (57%). Rats that received ECs in combination with MP had significantly more axons than all other lesioned rats up to 13 mm. Successful DFR performance was significantly higher in rats with EC transplants, both without (72%) and with (78%) MP, compared with other lesioned rats. These data confirm previous reports that ECs promote axonal regeneration and functional recovery after spinal cord lesions. In addition, this research provides evidence that, when used in combination, MP and ECs improve axonal regrowth up to 13 mm caudal to the lesion at 6 weeks after injury.
Figures


Similar articles
-
Effect of combined treatment with methylprednisolone and soluble Nogo-66 receptor after rat spinal cord injury.Eur J Neurosci. 2005 Aug;22(3):587-94. doi: 10.1111/j.1460-9568.2005.04241.x. Eur J Neurosci. 2005. PMID: 16101740 Free PMC article.
-
Chronic transplantation of olfactory ensheathing cells promotes partial recovery after complete spinal cord transection in the rat.Glia. 2007 Feb;55(3):303-11. doi: 10.1002/glia.20457. Glia. 2007. PMID: 17096411
-
Functional repair of the corticospinal tract by delayed transplantation of olfactory ensheathing cells in adult rats.J Neurosci. 2003 Oct 15;23(28):9428-34. doi: 10.1523/JNEUROSCI.23-28-09428.2003. J Neurosci. 2003. PMID: 14561871 Free PMC article.
-
Transplants and neurotrophic factors increase regeneration and recovery of function after spinal cord injury.Prog Brain Res. 2002;137:257-73. doi: 10.1016/s0079-6123(02)37020-1. Prog Brain Res. 2002. PMID: 12440372 Review.
-
Olfactory ensheathing cells: bridging the gap in spinal cord injury.Neurosurgery. 2000 Nov;47(5):1057-69. doi: 10.1097/00006123-200011000-00006. Neurosurgery. 2000. PMID: 11063098 Review.
Cited by
-
Chemotactic TEG3 Cells' Guiding Platforms Based on PLA Fibers Functionalized With the SDF-1α/CXCL12 Chemokine for Neural Regeneration Therapy.Front Bioeng Biotechnol. 2021 Mar 22;9:627805. doi: 10.3389/fbioe.2021.627805. eCollection 2021. Front Bioeng Biotechnol. 2021. PMID: 33829009 Free PMC article.
-
Taking a bite out of spinal cord injury: do dental stem cells have the teeth for it?Cell Mol Life Sci. 2016 Apr;73(7):1413-37. doi: 10.1007/s00018-015-2126-5. Epub 2016 Jan 14. Cell Mol Life Sci. 2016. PMID: 26768693 Free PMC article. Review.
-
Transplantation of canine olfactory ensheathing cells producing chondroitinase ABC promotes chondroitin sulphate proteoglycan digestion and axonal sprouting following spinal cord injury.PLoS One. 2017 Dec 11;12(12):e0188967. doi: 10.1371/journal.pone.0188967. eCollection 2017. PLoS One. 2017. PMID: 29228020 Free PMC article.
-
Cellular and paracellular transplants for spinal cord injury: a review of the literature.Childs Nerv Syst. 2011 Feb;27(2):237-43. doi: 10.1007/s00381-010-1312-x. Epub 2010 Oct 23. Childs Nerv Syst. 2011. PMID: 20972681 Review.
-
Impact of Dual Cell Co-culture and Cell-conditioned Media on Yield and Function of a Human Olfactory Cell Line for Regenerative Medicine.Bioengineering (Basel). 2020 Apr 12;7(2):37. doi: 10.3390/bioengineering7020037. Bioengineering (Basel). 2020. PMID: 32290611 Free PMC article.
References
-
- Almawi WY, Hadro ET, Strom TB. Evidence that glucocorticosteroid-mediated immunosuppressive effects do not involve altering second messenger function. Transplantation. 1991;52:133–140. - PubMed
-
- Bartolomei JC, Greer CA. Olfactory ensheathing cells: bridging the gap in spinal cord injury. Neurosurgery. 2000;47:1057–1069. - PubMed
-
- Becker J, Grasso RJ. Suppression of phagocytosis by dexamethasone in macrophage cultures: inability of arachidonic acid, indomethacin, and nordihydroguaiaretic acid to reverse the inhibitory response mediated by a steroid-inducible factor. Int J Immunopharmacol. 1985;7:839–847. - PubMed
-
- Bernstein-Goral H, Bregman BS. Axotomized rubrospinal neurons rescued by fetal spinal cord transplants maintain axon collaterals to rostral CNS targets. Exp Neurol. 1997;148:13–25. - PubMed
-
- Bracken MB, Collins WF, Freeman DF, Shepard MJ, Wagner FW, Silten RM, Hellenbrand KG, Ransohoff J, Hunt WE, Perot PL, Jr, Grossman RG, Green BA, Eisenberg HM, Rifkinson N, Goodman JH, Meagher JN, Fischer B, Clifton GL, Flamm ES, Rawe SE. Efficacy of methylprednisolone in acute spinal cord injury. JAMA. 1984;251:45–52. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
