Active site geometry of oxalate decarboxylase from Flammulina velutipes: Role of histidine-coordinated manganese in substrate recognition
- PMID: 12192069
- PMCID: PMC2373591
- DOI: 10.1110/ps.0206802
Active site geometry of oxalate decarboxylase from Flammulina velutipes: Role of histidine-coordinated manganese in substrate recognition
Abstract
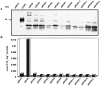
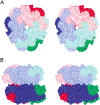
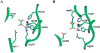
Oxalate decarboxylase (OXDC) from the wood-rotting fungus Flammulina velutipes, which catalyzes the conversion of oxalate to formic acid and CO(2) in a single-step reaction, is a duplicated double-domain germin family enzyme. It has agricultural as well as therapeutic importance. We reported earlier the purification and molecular cloning of OXDC. Knowledge-based modeling of the enzyme reveals a beta-barrel core in each of the two domains organized in the hexameric state. A cluster of three histidines suitably juxtaposed to coordinate a divalent metal ion exists in both the domains. Involvement of the two histidine clusters in the catalytic mechanism of the enzyme, possibly through coordination of a metal cofactor, has been hypothesized because all histidine knockout mutants showed total loss of decarboxylase activity. The atomic absorption spectroscopy analysis showed that OXDC contains Mn(2+) at up to 2.5 atoms per subunit. Docking of the oxalate in the active site indicates a similar electrostatic environment around the substrate-binding site in the two domains. We suggest that the histidine coordinated manganese is critical for substrate recognition and is directly involved in the catalysis of the enzyme.
Figures

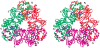


Similar articles
-
A calmodulin like EF hand protein positively regulates oxalate decarboxylase expression by interacting with E-box elements of the promoter.Sci Rep. 2015 Oct 12;5:14578. doi: 10.1038/srep14578. Sci Rep. 2015. PMID: 26455820 Free PMC article.
-
Cloning and characterization of the 5'-flanking region of the oxalate decarboxylase gene from Flammulina velutipes.Biochem J. 2002 Oct 1;367(Pt 1):67-75. doi: 10.1042/BJ20011573. Biochem J. 2002. PMID: 12020349 Free PMC article.
-
Substrate Binding Mode and Molecular Basis of a Specificity Switch in Oxalate Decarboxylase.Biochemistry. 2016 Apr 12;55(14):2163-73. doi: 10.1021/acs.biochem.6b00043. Epub 2016 Apr 4. Biochemistry. 2016. PMID: 27014926 Free PMC article.
-
Oxalate decarboxylase: biotechnological update and prevalence of the enzyme in filamentous fungi.Appl Microbiol Biotechnol. 2010 Jul;87(3):801-14. doi: 10.1007/s00253-010-2650-z. Epub 2010 May 13. Appl Microbiol Biotechnol. 2010. PMID: 20464388 Review.
-
Biological functions controlled by manganese redox changes in mononuclear Mn-dependent enzymes.Essays Biochem. 2017 May 9;61(2):259-270. doi: 10.1042/EBC20160070. Print 2017 May 9. Essays Biochem. 2017. PMID: 28487402 Review.
Cited by
-
Characterization of peanut germin-like proteins, AhGLPs in plant development and defense.PLoS One. 2013 Apr 23;8(4):e61722. doi: 10.1371/journal.pone.0061722. Print 2013. PLoS One. 2013. PMID: 23626720 Free PMC article.
-
Reduction of oxalate levels in tomato fruit and consequent metabolic remodeling following overexpression of a fungal oxalate decarboxylase.Plant Physiol. 2013 May;162(1):364-78. doi: 10.1104/pp.112.209197. Epub 2013 Mar 12. Plant Physiol. 2013. PMID: 23482874 Free PMC article.
-
A calmodulin like EF hand protein positively regulates oxalate decarboxylase expression by interacting with E-box elements of the promoter.Sci Rep. 2015 Oct 12;5:14578. doi: 10.1038/srep14578. Sci Rep. 2015. PMID: 26455820 Free PMC article.
-
Role of oxalic acid in fungal and bacterial metabolism and its biotechnological potential.World J Microbiol Biotechnol. 2024 Apr 25;40(6):178. doi: 10.1007/s11274-024-03973-5. World J Microbiol Biotechnol. 2024. PMID: 38662173 Free PMC article. Review.
-
Improving nutritional quality and fungal tolerance in soya bean and grass pea by expressing an oxalate decarboxylase.Plant Biotechnol J. 2016 Jun;14(6):1394-405. doi: 10.1111/pbi.12503. Epub 2016 Jan 22. Plant Biotechnol J. 2016. PMID: 26798990 Free PMC article.
References
-
- Altschul, S.F., Gish, W., Miller, W., Myers, E.W., and Lipman, D.J. 1990. Basic local alignment search tool. J. Mol. Biol. 215 403–410. - PubMed
-
- Banci, L., Bertini, I., Pozzo, L.D., Conte, R.D., and Tien, M. 1998. Monitoring the role of oxalate in manganese peroxidase. Biochemistry 37 9009–9015. - PubMed
-
- Baumlein, H., Braun, H., Kakhovskaya, I.A., and Shutov, A.D. 1995. Seed storage proteins of spermatophytes share a common ancestor with desiccation proteins of fungi. J. Mol. Evol. 41 1070–1075. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

