Novel adeno-associated viruses from rhesus monkeys as vectors for human gene therapy
- PMID: 12192090
- PMCID: PMC129358
- DOI: 10.1073/pnas.182412299
Novel adeno-associated viruses from rhesus monkeys as vectors for human gene therapy
Abstract
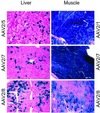
Tissues from rhesus monkeys were screened by PCR for the presence of sequences homologous to known adeno-associated virus (AAV) serotypes 1-6. DNA spanning entire rep-cap ORFs from two novel AAVs, called AAV7 and AAV8, were isolated. Sequence comparisons among these and previously described AAVs revealed the greatest divergence in capsid proteins. AAV7 and AAV8 were not neutralized by heterologous antisera raised to the other serotypes. Neutralizing antibodies to AAV7 and AAV8 were rare in human serum and, when present, were low in activity. Vectors formed with capsids from AAV7 and AAV8 were generated by using rep and inverted terminal repeats (ITRs) from AAV2 and were compared with similarly constructed vectors made from capsids of AAV1, AAV2, and AAV5. Murine models of skeletal muscle and liver-directed gene transfer were used to evaluate relative vector performance. AAV7 vectors demonstrated efficiencies of transgene expression in skeletal muscle equivalent to that observed with AAV1, the most efficient known serotype for this application. In liver, transgene expression was 10- to 100-fold higher with AAV8 than observed with other serotypes. This improved efficiency correlated with increased persistence of vector DNA and higher number of transduced hepatocytes. The efficiency of AAV8 vector for liver-directed gene transfer of factor IX was not impacted by preimmunization with the other AAV serotypes. Vectors based on these novel, nonhuman primate AAVs should be considered for human gene therapy because of low reactivity to antibodies directed to human AAVs and because gene transfer efficiency in muscle was similar to that obtained with the best known serotype, whereas, in liver, gene transfer was substantially higher than previously described.
Figures


References
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

