An external site controls closing of the epithelial Na+ channel ENaC
- PMID: 12205178
- PMCID: PMC2290510
- DOI: 10.1113/jphysiol.2002.022020
An external site controls closing of the epithelial Na+ channel ENaC
Abstract
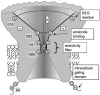
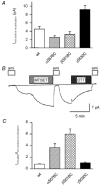
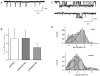
Members of the ENaC/degenerin family of ion channels include the epithelial sodium channel (ENaC), acid-sensing ion channels (ASICs) and the nematode Caenorhabditis elegans degenerins. These channels are activated by a variety of stimuli such as ligands (ASICs) and mechanical forces (degenerins), or otherwise are constitutively active (ENaC). Despite their functional heterogeneity, these channels might share common basic mechanisms for gating. Mutations of a conserved residue in the extracellular loop, namely the 'degenerin site' activate all members of the ENaC/degenerin family. Chemical modification of a cysteine introduced in the degenerin site of rat ENaC (betaS518C) by the sulfhydryl reagents MTSET or MTSEA, results in a approximately 3-fold increase in the open probability. This effect is due to an 8-fold shortening of channel closed times and an increase in the number of long openings. In contrast to the intracellular gating domain in the N-terminus which is critical for channel opening, the intact extracellular degenerin site is necessary for normal channel closing, as illustrated by our observation that modification of betaS518C destabilises the channel closed state. The modification by the sulfhydryl reagents is state- and size-dependent consistent with a conformational change of the degenerin site during channel opening and closing. We propose that the intracellular and extracellular modulatory sites act on a common channel gate and control the activity of ENaC at the cell surface.
Figures





Similar articles
-
Degenerin sites mediate proton activation of deltabetagamma-epithelial sodium channel.J Biol Chem. 2004 Jun 25;279(26):26939-47. doi: 10.1074/jbc.M401143200. Epub 2004 Apr 14. J Biol Chem. 2004. PMID: 15084585
-
Epithelial sodium channel pore region. structure and role in gating.J Biol Chem. 2001 Jan 12;276(2):1326-34. doi: 10.1074/jbc.M008117200. J Biol Chem. 2001. PMID: 11022046
-
A mutation causing pseudohypoaldosteronism type 1 identifies a conserved glycine that is involved in the gating of the epithelial sodium channel.EMBO J. 1997 Mar 3;16(5):899-907. doi: 10.1093/emboj/16.5.899. EMBO J. 1997. PMID: 9118951 Free PMC article.
-
Insight toward epithelial Na+ channel mechanism revealed by the acid-sensing ion channel 1 structure.IUBMB Life. 2008 Sep;60(9):620-8. doi: 10.1002/iub.89. IUBMB Life. 2008. PMID: 18459164 Review.
-
Epithelial sodium channel: a ligand-gated channel?Nephron Physiol. 2004;96(2):p37-41. doi: 10.1159/000076406. Nephron Physiol. 2004. PMID: 14988660 Review.
Cited by
-
Thumb domains of the three epithelial Na+ channel subunits have distinct functions.J Biol Chem. 2018 Nov 9;293(45):17582-17592. doi: 10.1074/jbc.RA118.003618. Epub 2018 Sep 18. J Biol Chem. 2018. PMID: 30228189 Free PMC article.
-
ENaC structure and function in the wake of a resolved structure of a family member.Am J Physiol Renal Physiol. 2011 Oct;301(4):F684-96. doi: 10.1152/ajprenal.00259.2011. Epub 2011 Jul 13. Am J Physiol Renal Physiol. 2011. PMID: 21753073 Free PMC article. Review.
-
Benzamil sensitive ion channels contribute to volume regulation in canine chondrocytes.Br J Pharmacol. 2013 Apr;168(7):1584-96. doi: 10.1111/j.1476-5381.2012.02185.x. Br J Pharmacol. 2013. PMID: 22928819 Free PMC article.
-
The delta-subunit of the epithelial sodium channel (ENaC) enhances channel activity and alters proteolytic ENaC activation.J Biol Chem. 2009 Oct 16;284(42):29024-40. doi: 10.1074/jbc.M109.018945. Epub 2009 Aug 28. J Biol Chem. 2009. PMID: 19717556 Free PMC article.
-
SECS, drugs, and Rac1&Rho: regulation of EnNaC in vascular endothelial cells.Pflugers Arch. 2025 Jul;477(7):977-992. doi: 10.1007/s00424-025-03093-5. Epub 2025 May 22. Pflugers Arch. 2025. PMID: 40402207 Free PMC article.
References
-
- Adams CM, Snyder PM, Price MP, Welsh MJ. Protons activate brain Na+ channel 1 by inducing a conformational change that exposes a residue associated with neurodegeneration. Journal of Biological Chemistry. 1998b;273:30204–30207. - PubMed
-
- Adams CM, Snyder PM, Welsh MJ. Paradoxical stimulation of a DEG/ENaC channel by amiloride. Journal of Biological Chemistry. 1999;274:15500–15504. - PubMed
-
- Champigny G, Voilley N, Waldmann R, Lazdunski M. Mutations causing neurodegeneration in Caenorhabditis elegans drastically alter the pH sensitivity and inactivation of the mammalian H+-gated Na+ channel MDEG1. Journal of Biological Chemistry. 1998;273:15418–15422. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

