Mechanisms of acid-induced activation of airway afferent nerve fibres in guinea-pig
- PMID: 12205192
- PMCID: PMC2290522
- DOI: 10.1113/jphysiol.2002.022848
Mechanisms of acid-induced activation of airway afferent nerve fibres in guinea-pig
Abstract
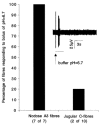
The mechanisms underlying the response of airway afferent nerves to low pH were investigated in an isolated guinea-pig airway nerve preparation. Extracellular recordings were made from single jugular or nodose vagal ganglion neurons that projected their sensory fibers into the airways. The airway tissue containing the mechanically sensitive receptive fields was exposed into acidic solutions. Rapid and transient (approximately 3 s) administration of 1 mM citric acid to the receptive field consistently induced action potential discharge in nociceptive C-fibers (41/44) and nodose Adelta fibres (29/30) that are rapidly adapting low threshold mechanosensors (RAR-like fibres). In contrast, citric acid activated only 8/17 high threshold mechanosensitive jugular Adelta fibres. The RAR-like fibres were slightly more sensitive than C-fibres to acidic solutions (pH threshold > 6.7). The RAR-like fibres response to the approximately 3 s acid treatment was not affected by a vanilloid receptor 1 (VR1) antagonist, capsazepine (10 microM), and was rapidly inactivating (action potential discharge terminated before the acid administration was completed). Gradual reduction of pH did not activate the RAR-like fibres even when the pH was reduced to approximately 5.0. The C-fibres responded to the gradual reduction of pH with persistent action potential discharge that was nearly abolished by capsazepine (10 microM) and inhibited by over 70 % with another VR1 antagonist iodo-resiniferatoxin (1 microM). In contrast the C-fibre response to the transient approximately 3 s exposure to pH approximately 5.0 was not affected by the VR1 antagonists. We conclude that activation of guinea-pig airway afferents by low pH is mediated by both slowly and rapidly inactivating mechanisms. We hypothesize that the slowly inactivating mechanism, present in C-fibres but not in RAR-like fibres, is mediated by VR1. The rapidly inactivating mechanism acts independently of VR1, has characteristics similar to acid sensing ion channels (ASICs) and is found in the airway terminals of both C-fibres and RAR-like fibres.
Figures



Similar articles
-
Effect of olvanil and anandamide on vagal C-fiber subtypes in guinea pig lung.Br J Pharmacol. 2005 Oct;146(4):596-603. doi: 10.1038/sj.bjp.0706339. Br J Pharmacol. 2005. PMID: 16056239 Free PMC article.
-
Physiology and plasticity of putative cough fibres in the Guinea pig.Pulm Pharmacol Ther. 2002;15(3):193-8. doi: 10.1006/pupt.2002.0362. Pulm Pharmacol Ther. 2002. PMID: 12099763 Review.
-
Effect of extracellular calcium on excitability of guinea pig airway vagal afferent nerves.J Neurophysiol. 2003 Mar;89(3):1196-204. doi: 10.1152/jn.00553.2002. J Neurophysiol. 2003. PMID: 12626607
-
Subtypes of vagal afferent C-fibres in guinea-pig lungs.J Physiol. 2004 May 1;556(Pt 3):905-17. doi: 10.1113/jphysiol.2003.060079. Epub 2004 Feb 20. J Physiol. 2004. PMID: 14978204 Free PMC article.
-
Functional morphology and physiological properties of bronchopulmonary C-fiber afferents.Anat Rec A Discov Mol Cell Evol Biol. 2003 Jan;270(1):17-24. doi: 10.1002/ar.a.10005. Anat Rec A Discov Mol Cell Evol Biol. 2003. PMID: 12494486 Review.
Cited by
-
Hypersensitivity of pulmonary chemosensitive neurons induced by activation of protease-activated receptor-2 in rats.J Physiol. 2006 Aug 1;574(Pt 3):867-76. doi: 10.1113/jphysiol.2006.110312. Epub 2006 May 18. J Physiol. 2006. PMID: 16709636 Free PMC article.
-
Sensory detection and responses to toxic gases: mechanisms, health effects, and countermeasures.Proc Am Thorac Soc. 2010 Jul;7(4):269-77. doi: 10.1513/pats.201001-004SM. Proc Am Thorac Soc. 2010. PMID: 20601631 Free PMC article. Review.
-
Effects of a high-salt diet on TRPV-1-dependent renal nerve activity in Dahl salt-sensitive rats.Am J Nephrol. 2010;32(3):194-200. doi: 10.1159/000316528. Epub 2010 Jul 15. Am J Nephrol. 2010. PMID: 20639627 Free PMC article.
-
Differential effects of airway afferent nerve subtypes on cough and respiration in anesthetized guinea pigs.Am J Physiol Regul Integr Comp Physiol. 2008 Nov;295(5):R1572-84. doi: 10.1152/ajpregu.90382.2008. Epub 2008 Sep 3. Am J Physiol Regul Integr Comp Physiol. 2008. PMID: 18768768 Free PMC article.
-
Cough and gastroesophageal reflux: insights from animal models.Pulm Pharmacol Ther. 2009 Apr;22(2):130-4. doi: 10.1016/j.pupt.2008.12.017. Epub 2008 Dec 31. Pulm Pharmacol Ther. 2009. PMID: 19138751 Free PMC article. Review.
References
-
- Andersson SE, Lexmuller K, Johansson A, Ekstrom GM. Tissue and intracellular pH in normal periarticular soft tissue and during different phases of antigen induced arthritis in the rat. Journal of Rheumatology. 1999;26:2018–2024. - PubMed
-
- Berger KI, Ayappa I, Sorkin IB, Norman RG, Rapoport DM, Goldring RM. CO2 homeostasis during periodic breathing in obstructive sleep apnea. Journal of Applied Physiology. 2000;88:257–264. - PubMed
-
- Bolser DC, AziZ SM, Chapman RW. Ruthenium Red decreases capsaicin and citric acid-induced cough in guinea pigs. Neuroscience Letters. 1991;126:131–133. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

