Bni5p, a septin-interacting protein, is required for normal septin function and cytokinesis in Saccharomyces cerevisiae
- PMID: 12215547
- PMCID: PMC134035
- DOI: 10.1128/MCB.22.19.6906-6920.2002
Bni5p, a septin-interacting protein, is required for normal septin function and cytokinesis in Saccharomyces cerevisiae
Abstract
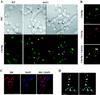
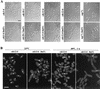
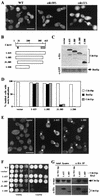
In the budding yeast Saccharomyces cerevisiae, the Cdc3p, Cdc10p, Cdc11p, Cdc12p, and Sep7p/Shs1p septins assemble early in the cell cycle in a ring that marks the future cytokinetic site. The septins appear to be major structural components of a set of filaments at the mother-bud neck and function as a scaffold for recruiting proteins involved in cytokinesis and other processes. We isolated a novel gene, BNI5, as a dosage suppressor of the cdc12-6 growth defect. Overexpression of BNI5 also suppressed the growth defects of cdc10-1, cdc11-6, and sep7Delta strains. Loss of BNI5 resulted in a cytokinesis defect, as evidenced by the formation of connected cells with shared cytoplasms, and deletion of BNI5 in a cdc3-6, cdc10-1, cdc11-6, cdc12-6, or sep7Delta mutant strain resulted in enhanced defects in septin localization and cytokinesis. Bni5p localizes to the mother-bud neck in a septin-dependent manner shortly after bud emergence and disappears from the neck approximately 2 to 3 min before spindle disassembly. Two-hybrid, in vitro binding, and protein-localization studies suggest that Bni5p interacts with the N-terminal domain of Cdc11p, which also appears to be sufficient for the localization of Cdc11p, its interaction with other septins, and other critical aspects of its function. Our data suggest that the Bni5p-septin interaction is important for septin ring stability and function, which is in turn critical for normal cytokinesis.
Figures





Similar articles
-
Protein-protein interactions governing septin heteropentamer assembly and septin filament organization in Saccharomyces cerevisiae.Mol Biol Cell. 2004 Oct;15(10):4568-83. doi: 10.1091/mbc.e04-04-0330. Epub 2004 Jul 28. Mol Biol Cell. 2004. PMID: 15282341 Free PMC article.
-
Polymerization of purified yeast septins: evidence that organized filament arrays may not be required for septin function.J Cell Biol. 1998 Nov 2;143(3):737-49. doi: 10.1083/jcb.143.3.737. J Cell Biol. 1998. PMID: 9813094 Free PMC article.
-
Phosphorylation-dependent septin interaction of Bni5 is important for cytokinesis.J Microbiol. 2007 Jun;45(3):227-33. J Microbiol. 2007. PMID: 17618228
-
Regulation of septin organization and function in yeast.Trends Cell Biol. 2003 Aug;13(8):403-9. doi: 10.1016/s0962-8924(03)00151-x. Trends Cell Biol. 2003. PMID: 12888292 Review.
-
Some assembly required: yeast septins provide the instruction manual.Trends Cell Biol. 2005 Aug;15(8):414-24. doi: 10.1016/j.tcb.2005.06.007. Trends Cell Biol. 2005. PMID: 16009555 Free PMC article. Review.
Cited by
-
Septin Assembly and Remodeling at the Cell Division Site During the Cell Cycle.Front Cell Dev Biol. 2021 Nov 25;9:793920. doi: 10.3389/fcell.2021.793920. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34901034 Free PMC article. Review.
-
Budding yeast Dma1 and Dma2 participate in regulation of Swe1 levels and localization.Mol Biol Cell. 2011 Jul 1;22(13):2185-97. doi: 10.1091/mbc.E11-02-0127. Epub 2011 May 11. Mol Biol Cell. 2011. PMID: 21562220 Free PMC article.
-
Connecting cell polarity signals to the cytokinetic machinery in yeast and metazoan cells.Cell Cycle. 2021 Jan;20(1):1-10. doi: 10.1080/15384101.2020.1864941. Epub 2021 Jan 5. Cell Cycle. 2021. PMID: 33397181 Free PMC article. Review.
-
Bni5 tethers myosin-II to septins to enhance retrograde actin flow and the robustness of cytokinesis.bioRxiv [Preprint]. 2023 Nov 8:2023.11.07.566094. doi: 10.1101/2023.11.07.566094. bioRxiv. 2023. PMID: 37986946 Free PMC article. Preprint.
-
Septin-Associated Protein Kinases in the Yeast Saccharomyces cerevisiae.Front Cell Dev Biol. 2016 Nov 1;4:119. doi: 10.3389/fcell.2016.00119. eCollection 2016. Front Cell Dev Biol. 2016. PMID: 27847804 Free PMC article. Review.
References
-
- Ausubel, F. M., R. Brent, R. E. Kingston, D. D. Moore, J. G. Seidman, J. A. Smith, and K. Struhl. 1995. Current protocols in molecular biology. John Wiley and Sons, Ltd., New York, N.Y.
-
- Barral, Y., V. Mermall, M. S. Mooseker, and M. Snyder. 2000. Compartmentalization of the cell cortex by septins is required for maintenance of cell polarity in yeast. Mol. Cell 5:841-851. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
