Cytoarchitecture of the human cerebral cortex: MR microscopy of excised specimens at 9.4 Tesla
- PMID: 12223371
- PMCID: PMC7976254
Cytoarchitecture of the human cerebral cortex: MR microscopy of excised specimens at 9.4 Tesla
Abstract
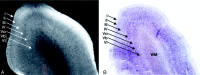
Background and purpose: The laminar patterns displayed by MR microscopy (MRM) form one basis for the classification of the cytoarchitectonic areas (Brodmann areas). It is plausible that in the future MRM may depict Brodmann areas directly, and not only by inference from gross anatomic location. Our purpose was to depict the laminar cytoarchitecture of excised, formalin-fixed specimens of human cerebral cortex by use of 9.4-T MR and to correlate MR images with histologic stains of the same sections.
Methods: Formalin-fixed samples of human sensory isocortex (calcarine, Heschl's, and somatosensory cortices), motor isocortex (hand motor area of M1), polar isocortex (frontal pole), allocortex (hippocampal formation), and transitional periallocortex (retrosplenial cortex) were studied by MRM at 9.4 T with intermediate-weighted pulse sequences for a total overnight acquisition time of 14 hours 17 minutes for each specimen. The same samples were then histologically analyzed to confirm the MR identification of the cortical layers. Curves representing the change in MR signal intensity across the cortex were generated to display the signal intensity profiles for each type of cortex.
Results: High-field-strength MR imaging at a spatial resolution of 78 x 78 x 500 micro m resolves the horizontal lamination of isocortex, allocortex, and periallocortex and displays specific intracortical structures such as the external band of Baillarger. The signal intensity profiles demonstrate the greatest hypointensity at the sites of maximum myelin concentration and maximum cell density and show gradations of signal intensity inversely proportional to varying cell density.
Conclusion: MRM at 9.4 T depicts important aspects of the cytoarchitecture of normal formalin-fixed human cortex.
Figures




References
-
- Vandersteen M, Beuls E, Gelan J, Adriaensens P, Vanormelingen L, Palmers Y, Freling G. High field magnetic resonance imaging of normal and pathologic medulla oblongata. Anat Rec 1994;238:277–286 - PubMed
-
- Benveniste H, Katie K, Zhang L, Johnson GA. Magnetic resonance microscopy of the C57BL mouse brain. NeuroImage 2000;11:601–611 - PubMed
-
- Burgess RE, Yu Y, Christoforidis GA, Bourkes EC, Chakeres DW, Dimitri S, Kangarlu A, Abduljalil AM, Robitaille PL. Human leptomeningeal and cortical vascular anatomy of the cerebral cortex at 8 T. J Comput Assis Tomogr 1999;23:850–856 - PubMed
-
- Carpenter MB, Sutin J. The cerebral cortex Human Neuroanatomy Baltimore: Williams & Wilkins;1983;643–705
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
