Role of presynaptic muscarinic and GABA(B) receptors in spinal glutamate release and cholinergic analgesia in rats
- PMID: 12231640
- PMCID: PMC2290545
- DOI: 10.1113/jphysiol.2002.020644
Role of presynaptic muscarinic and GABA(B) receptors in spinal glutamate release and cholinergic analgesia in rats
Abstract
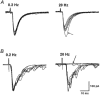
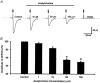
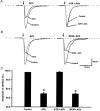
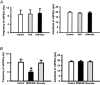
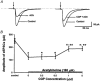
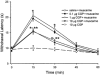
Spinally administered muscarinic receptor agonists or acetylcholinesterase inhibitors can produce effective pain relief. However, the analgesic mechanisms and the site of actions of cholinergic agents in the spinal cord are not fully understood. In this study, we investigated the mechanisms underlying cholinergic presynaptic regulation of glutamate release onto spinal dorsal horn neurons. The role of spinal GABA(B) receptors in the antinociceptive action of muscarine was also determined. Whole-cell voltage-clamp recordings were performed on visualized dorsal horn neurons in the lamina II in the spinal cord slice preparation of rats. The miniature excitatory postsynaptic currents (mEPSCs) and miniature inhibitory postsynaptic currents (mIPSCs) were recorded in the presence of tetrodotoxin. The evoked EPSCs (eEPSCs) were obtained by electrical stimulation of the dorsal root entry zone or the attached dorsal root. Nociception in rats was measured using a radiant heat stimulus and the effect of intrathecal administration of drugs tested. Acetylcholine (10-100 microM) reduced the amplitude of monosynaptic eEPSCs in a concentration-dependent manner. Acetylcholine also significantly decreased the frequency of non-NMDA receptor-mediated mEPSCs, which was antagonized by atropine but not mecamylamine. The frequency of GABA(A) receptor-mediated mIPSCs was significantly increased by acetylcholine and this excitatory effect was abolished by atropine. Existence of presynaptic M(2) muscarinic receptors in the spinal dorsal horn was further demonstrated by immunocytochemistry staining and dorsal rhizotomy. CGP55845, a GABA(B) receptor antagonist, significantly attenuated the inhibitory effect of acetylcholine on the frequency of mEPSCs and the amplitude of monosynaptic eEPSCs in lamina II neurons. Furthermore, the antinociceptive action produced by intrathecal muscarine was significantly reduced by CGP55845 pretreatment in rats. Therefore, data from this integrated study provide new information that acetylcholine inhibits the glutamatergic synaptic input to lamina II neurons through presynaptic muscarinic receptors. Inhibition of glutamate release onto lamina II neurons by presynaptic muscarinic and GABA(B) heteroreceptors in the spinal cord probably contributes to the antinociceptive action of cholinergic agents.
Figures


Similar articles
-
Dynamic regulation of glycinergic input to spinal dorsal horn neurones by muscarinic receptor subtypes in rats.J Physiol. 2006 Mar 1;571(Pt 2):403-13. doi: 10.1113/jphysiol.2005.102905. Epub 2006 Jan 12. J Physiol. 2006. PMID: 16410279 Free PMC article.
-
Cholinergic modulation of primary afferent glutamatergic transmission in rat medullary dorsal horn neurons.Neuropharmacology. 2013 Dec;75:295-303. doi: 10.1016/j.neuropharm.2013.07.030. Epub 2013 Aug 13. Neuropharmacology. 2013. PMID: 23954675
-
Regulation of glutamate release from primary afferents and interneurons in the spinal cord by muscarinic receptor subtypes.J Neurophysiol. 2007 Jan;97(1):102-9. doi: 10.1152/jn.00586.2006. Epub 2006 Oct 18. J Neurophysiol. 2007. PMID: 17050831
-
Pre- and postsynaptic inhibitory control in the spinal cord dorsal horn.Ann N Y Acad Sci. 2013 Mar;1279:90-6. doi: 10.1111/nyas.12056. Ann N Y Acad Sci. 2013. PMID: 23531006 Free PMC article. Review.
-
Muscarinic-mediated analgesia.Life Sci. 1999;64(6-7):549-54. doi: 10.1016/s0024-3205(98)00600-6. Life Sci. 1999. PMID: 10069522 Review.
Cited by
-
Muscarinic control of AMPA receptor responsiveness in mouse spinal cord motoneurons.J Physiol. 2012 Oct 1;590(19):4663-71. doi: 10.1113/jphysiol.2012.238444. Epub 2012 Aug 13. J Physiol. 2012. PMID: 22890702 Free PMC article.
-
Acetyl-L-carnitine in neuropathic pain: experimental data.CNS Drugs. 2007;21 Suppl 1:31-8; discussion 45-6. doi: 10.2165/00023210-200721001-00005. CNS Drugs. 2007. PMID: 17696591 Review.
-
Tiam1-mediated maladaptive plasticity underlying morphine tolerance and hyperalgesia.Brain. 2024 Jul 5;147(7):2507-2521. doi: 10.1093/brain/awae106. Brain. 2024. PMID: 38577773 Free PMC article.
-
Role of M2, M3, and M4 muscarinic receptor subtypes in the spinal cholinergic control of nociception revealed using siRNA in rats.J Neurochem. 2009 Nov;111(4):1000-10. doi: 10.1111/j.1471-4159.2009.06396.x. Epub 2009 Sep 23. J Neurochem. 2009. PMID: 19780895 Free PMC article.
-
Cholinergic mechanisms in spinal locomotion-potential target for rehabilitation approaches.Front Neural Circuits. 2014 Nov 6;8:132. doi: 10.3389/fncir.2014.00132. eCollection 2014. Front Neural Circuits. 2014. PMID: 25414645 Free PMC article.
References
-
- Chen S-R, Eisenach JC, McCaslin PP, Pan H-L. Synergistic effect between intrathecal non-NMDA antagonist and gabapentin on allodynia induced by spinal nerve ligation in rats. Anesthesiology. 2000;92:500–506. - PubMed
-
- Chen SR, Pan HL. Spinal endogenous acetylcholine contributes to the analgesic effect of systemic morphine in rats. Anesthesiology. 2001;95:525–530. - PubMed
-
- Chery N, De Koninck Y. GABAB receptors are the first target of released GABA at lamina I inhibitory synapses in the adult rat spinal cord. Journal of Neurophysiology. 2000;84:1006–1011. - PubMed
-
- Cui JG, Meyerson BA, Sollevi A, Linderoth B. Effect of spinal cord stimulation on tactile hypersensitivity in mononeuropathic rats is potentiated by simultaneous GABAB and adenosine receptor activation. Neuroscience Letters. 1998;247:183–186. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

