Loss of enteric motor neurotransmission in the gastric fundus of Sl/Sl(d) mice
- PMID: 12231645
- PMCID: PMC2290528
- DOI: 10.1113/jphysiol.2002.021915
Loss of enteric motor neurotransmission in the gastric fundus of Sl/Sl(d) mice
Abstract
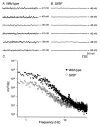
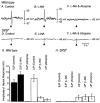
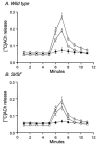
Studies of W/W(V) mice, which lack intramuscular interstitial cells of Cajal (IC-IM), have suggested that IC-IM act as mediators of enteric motor neurotransmission in the gastrointestinal tract. We have studied Sl/Sl(d) mice, which lack the ability to make membrane-bound stem cell factor, to determine the consequences of inappropriate stem cell factor expression on IC-IM populations and on enteric motor neurotransmission. IC-IM were found within the circular and longitudinal muscles of the gastric fundus of wild-type mice. IC-IM were intimately associated with motor nerve terminals and nerve varicosities formed synaptic structures with these cells. IC-IM were also connected with neighbouring smooth muscle cells via gap junctions. Immunohistochemistry and electron microscopy showed that IC-IM were absent from fundus muscles of Sl/Sl(d) mice, but the density of excitatory and inhibitory nerves was not significantly different than in wild-type muscles. Loss of IC-IM was associated with decreased membrane noise (unitary potentials) and significant reductions in post-junctional excitatory and inhibitory enteric nerve responses. Reductions in neural responses were not due to defects in smooth muscle cells as responses to exogenous ACh and K(+)-induced depolarization were normal in Sl/Sl(d) mice. Responses to neurally released ACh were revealed in Sl/Sl(d) mice by inhibiting ACh breakdown with the acetylcholinesterase inhibitor neostigmine. Inhibitory nerve stimulation elicited inhibitory junction potentials (IJPs) and relaxations in wild-type mice. IJPs were reduced in amplitude and relaxation responses were absent in Sl/Sl(d) mice. These observations suggest that membrane-bound stem cell factor is essential for development of IC-IM and that the close, synaptic-like relationship between nerve terminals and IC-IM may be the primary site of innervation by enteric motor neurons in gastric muscles.
Figures







References
-
- Daniel EE, Posey-Daniel V. Neuromuscular structures in opossum esophagus: role of interstitial cells of Cajal. American Journal of Physiology. 1984;246:G305–315. - PubMed
-
- Desai KM, Sessa WC, Vane JR. Involvement of nitric oxide in the reflex relaxation of the stomach to accommodate food or fluid. Nature. 1991;351:477–479. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

