Zmpste24 deficiency in mice causes spontaneous bone fractures, muscle weakness, and a prelamin A processing defect
- PMID: 12235369
- PMCID: PMC130584
- DOI: 10.1073/pnas.192460799
Zmpste24 deficiency in mice causes spontaneous bone fractures, muscle weakness, and a prelamin A processing defect
Abstract
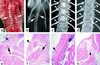
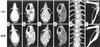
Zmpste24 is an integral membrane metalloproteinase of the endoplasmic reticulum. Biochemical studies of tissues from Zmpste24-deficient mice (Zmpste24(-/-)) have indicated a role for Zmpste24 in the processing of CAAX-type prenylated proteins. Here, we report the pathologic consequences of Zmpste24 deficiency in mice. Zmpste24(-/-) mice gain weight slowly, appear malnourished, and exhibit progressive hair loss. The most striking pathologic phenotype is multiple spontaneous bone fractures-akin to those occurring in mouse models of osteogenesis imperfecta. Cortical and trabecular bone volumes are significantly reduced in Zmpste24(-/-) mice. Zmpste24(-/-) mice also manifested muscle weakness in the lower and upper extremities, resembling mice lacking the farnesylated CAAX protein prelamin A. Prelamin A processing was defective both in fibroblasts lacking Zmpste24 and in fibroblasts lacking the CAAX carboxyl methyltransferase Icmt but was normal in fibroblasts lacking the CAAX endoprotease Rce1. Muscle weakness in Zmpste24(-/-) mice can be reasonably ascribed to defective processing of prelamin A, but the brittle bone phenotype suggests a broader role for Zmpste24 in mammalian biology.
Figures




References
-
- Boyartchuk V L, Ashby M N, Rine J. Science. 1997;275:1796–1800. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

