Naturally occurring substitutions in the P/V gene convert the noncytopathic paramyxovirus simian virus 5 into a virus that induces alpha/beta interferon synthesis and cell death
- PMID: 12239285
- PMCID: PMC136585
- DOI: 10.1128/jvi.76.20.10109-10121.2002
Naturally occurring substitutions in the P/V gene convert the noncytopathic paramyxovirus simian virus 5 into a virus that induces alpha/beta interferon synthesis and cell death
Abstract
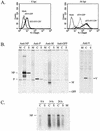
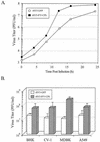
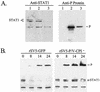
The V protein of the paramyxovirus simian virus 5 (SV5) is responsible for targeted degradation of STAT1 and the block in alpha/beta interferon (IFN-alpha/beta) signaling that occurs after SV5 infection of human cells. We have analyzed the growth properties of a recombinant SV5 that was engineered to be defective in targeting STAT1 degradation. A recombinant SV5 (rSV5-P/V-CPI-) was engineered to contain six naturally occurring P/V protein mutations, three of which have been shown in previous transfection experiments to disrupt the V-mediated block in IFN-alpha/beta signaling. In contrast to wild-type (WT) SV5, human cells infected with rSV5-P/V-CPI- had STAT1 levels similar to those in mock-infected cells. Cells infected with rSV5-P/V-CPI- were found to express higher-than-WT levels of viral proteins and mRNA, suggesting that the P/V mutations had disrupted the regulation of viral RNA synthesis. Despite the inability to target STAT1 for degradation, single-step growth assays showed that the rSV5-P/V-CPI- mutant virus grew better than WT SV5 in all cell lines tested. Unexpectedly, cells infected with rSV5-P/V-CPI- but not WT SV5 showed an activation of a reporter gene that was under control of the IFN-beta promoter. The secretion of IFN from cells infected with rSV5-P/V-CPI- but not WT SV5 was confirmed by a bioassay for IFN. The rSV5-P/V-CPI- mutant grew to higher titers than did WT rSV5 at early times in multistep growth assays. However, rSV5-P/V-CPI- growth quickly reached a final plateau while WT rSV5 continued to grow and produced a final titer higher than that of rSV5-P/V-CPI- by late times postinfection. In contrast to WT rSV5, infection of a variety of cell lines with rSV5-P/V-CPI- induced cell death pathways with characteristics of apoptosis. Our results confirm a role for the SV5 V protein in blocking IFN signaling but also suggest new roles for the P/V gene products in controlling viral gene expression, the induction of IFN-alpha/beta synthesis, and virus-induced apoptosis.
Figures





References
-
- Atreya, P. L., and S. Kulkarni. 1999. Respiratory syncytial virus A2 is resistant to the antiviral effects of type I interferons and human MxA. Virology 261:227-241. - PubMed
-
- Baumgartner, W. K., S. Krakowka, and B. Durchfeld. 1991. In vitro cytopathogenicity and in vivo virulence of two strains of canine parainfluenza virus. Vet. Pathol. 28:324-331. - PubMed
-
- Biron, C. A., and G. C. Sen. 2001. Interferons and other cytokines, p. 321-349. In D. Knipe, P. Howley, D. Griffin, et al. (ed.), Fields virology, 4th ed. Lippincott-Raven Publishers, Philadelphia, Pa.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

