Calpain as an effector of the Gq signaling pathway for inhibition of Wnt/beta -catenin-regulated cell proliferation
- PMID: 12239346
- PMCID: PMC130620
- DOI: 10.1073/pnas.202355799
Calpain as an effector of the Gq signaling pathway for inhibition of Wnt/beta -catenin-regulated cell proliferation
Abstract
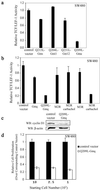
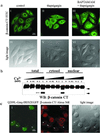
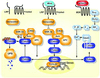
Signaling pathways interact to integrate and regulate information flow in evoking complex cellular responses. We have studied the mechanisms and consequences of interactions between the Gq and Wnt/beta-catenin pathways. In human colon carcinoma SW480 cells, activation of the Gq pathway inhibits beta-catenin signaling as determined by transcriptional reporter and cell proliferation assays. Ca(2+) release from internal stores results in nuclear export and calpain-mediated degradation of beta-catenin in the cytoplasm. Galphaq does not inhibit the effects of constitutively activated DeltaN-XTCF3-VP16 chimera in SW480 cells. Similarly, in HEK293 cells the Gq pathway suppresses beta-catenin-T cell factor/lymphocyte enhancer factor-1 transcriptional activity induced by Wnt/Frizzled interaction or glycogen synthase kinase-3beta-resistant beta-catenin, but not DeltaN-XTCF3-VP16. We conclude that Gq signaling promotes nuclear export and calpain-mediated degradation of beta-catenin, which therefore contributes to the inhibition of Wnt/beta-catenin pathway.
Figures


References
-
- Chen J, Iyengar R. Science. 1994;263:1278–1281. - PubMed
-
- Ram P T, Horvath C M, Iyengar R. Science. 2000;287:142–144. - PubMed
-
- Blitzer R D, Connor J H, Brown G P, Wong T, Shenolikar S, Iyengar R, Landau E M. Science. 1998;280:1940–1942. - PubMed
-
- Cowley S, Paterson H, Kemp P, Marshall C J. Cell. 1994;77:841–852. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

