Detection and identification of a soy protein component that cross-reacts with caseins from cow's milk
- PMID: 12296853
- PMCID: PMC1906491
- DOI: 10.1046/j.1365-2249.2002.t01-1-01935.x
Detection and identification of a soy protein component that cross-reacts with caseins from cow's milk
Abstract
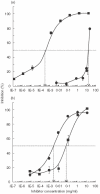
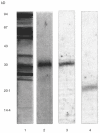
Soy-based formulas are the most employed cow's milk substitutes in the treatment of cow's milk allergy in our country. Since adverse reactions have been reported in allergic patients as a consequence of exposure to soy proteins, we have investigated the possible cross-reactivity between components from soybean and cow's milk. A cow's milk specific polyclonal antiserum and casein specific monoclonal antibodies were used in immunoblotting and competitive ELISA studies to identify a 30-kD component from soybean that cross-reacts with cow's milk caseins. Its IgE binding capacity was tested by EAST, employing sera from cow's milk allergic patients, not previously exposed to soy proteins. The 30 kD protein was isolated and partially sequenced. It is constituted by two polypeptides (A5 and B3) linked by a disulphide bond. The protein's capacity to bind to the different antibodies relies on the B3 poly-peptide. These results indicate that soy-based formula, which contains the A5-B3 glycinin molecule, could be involved in allergic reactions observed in cow's milk allergic patients exposed to soy-containing foods.
Figures




References
-
- Bock SA, Atkins FM. Patterns of food hypersensitivity during sixteen years of double-blind, placebo-controlled food challenges. J Pediatr. 1990;117:561–7. - PubMed
-
- Sampson HA. Food allergy. Part 1: immunopathogenesis and clinical disorders. J Allergy Clin Immunol. 1999;103:717–28. - PubMed
-
- Docena G, Fernandez R, Chirdo F, Fossati C. Identification of casein as the major allergenic and antigenic protein of cow’smilk. Allergy. 1996;51:412–6. - PubMed
-
- Restani P, Gaiaschi A, Plebani A, et al. Cross-reactivity between milk proteins from different animal species. Clin Exp Allergy. 1999;29:997–1004. - PubMed
-
- Gjesing B, Osterballe O, Schwartz B, Wahn U, Lowenstein H. Allergen-specific IgE antibodies against antigenic components in cow's milk and milk substitutes. Allergy. 1986;41:51–6. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

