A potassium ion channel is involved in cytokine production by activated human macrophages
- PMID: 12296855
- PMCID: PMC1906495
- DOI: 10.1046/j.1365-2249.2002.01965.x
A potassium ion channel is involved in cytokine production by activated human macrophages
Erratum in
- Clin Exp Immunol 2002 Dec;130(3):559-61
Abstract
Macrophages play an important role in immune and inflammatory responses, largely through secretion of bioactive molecule such as cytokines. While calcium is known to be an important regulator of this process, less is known about the role of other ions and the ion channels that regulate them. We have previously implicated an outwardly rectifying potassium channel (Kor) in this process and for this reason we have investigated the role of potassium (K+) and K+ channels in the regulation of tumour necrosis factor-alpha (TNF-alpha)and interleukin (IL)-8 production by activated human culture-derived macrophages. The effect of blockade of Kor is to inhibit phorbol myristate acetate (PMA)-induced cytokine production by translational or post-translational mechanisms, an effect that is duplicated by increasing extracellular K+. By contrast, the effects of K+ on LPS-stimulated cells are far more complex and are probably mediated through the change of osmolality and occur largely at the mRNA level. This data directly implicates K+, and its regulation through Kor, in early events following PMA stimulation of these cells.
Figures
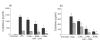
 , IL-8 (×100)
, IL-8 (×100)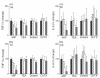
 , low; □, medium;
, low; □, medium; high. (c, d): ▪, PMA only;
high. (c, d): ▪, PMA only;  , low; □, medium;
, low; □, medium;  , high.
, high.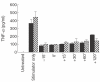
 , PMA (40 n
, PMA (40 n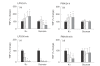
 , 20 mM; □, 60 mM;
, 20 mM; □, 60 mM;  , 90 mM. (b, d): ▪, PMA only;
, 90 mM. (b, d): ▪, PMA only;  ;, 20 mM; □, 60 mM;
;, 20 mM; □, 60 mM;  , 90 mM.
, 90 mM.
 4AP stimulator. (c, d): ▪, stimulator only;
4AP stimulator. (c, d): ▪, stimulator only;  K+/stimulator; □, sucrose/stimulator.
K+/stimulator; □, sucrose/stimulator.References
-
- Sweet MJ, Hume DA. Endotoxin signal transduction in macrophages. J Leukoc Biol. 1996;60:8–26. - PubMed
-
- Gordon S, Keshav S, Chung LP. Mononuclear phagocytes: tissue distribution and functional heterogeneity. Curr Opin Immunol. 1988;1:26–35. - PubMed
-
- Premack BA, Gardner P. Role of ion channels in lymphocytes. J Clin Immunol. 1991;11:225–38. - PubMed
-
- Sutro JB, Vayuvegula BS, Gupta S, et al. Voltage-sensitive ion channels in human B lymphocytes. Adv Exp Med Biol. 1989;254:113–22. - PubMed
-
- Amigorena S, Choquet D, Teillaud JL, et al. Ion channel blockers inhibit B cell activation at a precise stage of the G1 phase of the cell cycle. Possible involvement of K+ channels. J Immunol. 1990;144:2038–45. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

