Disruption of early events in thalamocortical tract formation in mice lacking the transcription factors Pax6 or Foxg1
- PMID: 12351726
- PMCID: PMC6218002
- DOI: 10.1523/JNEUROSCI.22-19-08523.2002
Disruption of early events in thalamocortical tract formation in mice lacking the transcription factors Pax6 or Foxg1
Abstract
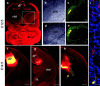
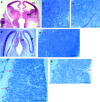
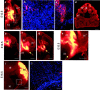
Early events in the formation of the thalamocortical tract remain poorly understood. Recent work has suggested that thalamocortical axons follow a path pioneered by transient thalamic afferents originating from the medial part of the ventral telencephalon. We studied the development of these transient afferents and the thalamocortical tract in mutant mice lacking transcription factors normally expressed in the dorsal thalamus or ventral telencephalon. Pax6 is expressed in the dorsal thalamus, but not in the medial part of the ventral telencephalon, and the thalamocortical tract fails to form in Pax6(-/-) embryos. We found that transient thalamic afferents from the ventral telencephalon do not form in Pax6(-/-) embryos; this may contribute to the failure of their thalamocortical development. The distribution of Pax6(-/-) cells in Pax6(-/-)<--> Pax6(+/+) chimeras supports conclusions drawn from forebrain marker gene expression that Pax6 is not required for the normal development of the medial part of the ventral telencephalon but is required in the dorsal thalamus. Failure of the transient afferent pathway to develop is therefore likely a cell nonautonomous defect reflecting primary defects in the thalamus. We then examined the formation of thalamic afferents and efferents in Foxg1(-/-) embryos, which lack recognizable ventral telencephalic structures. In these embryos thalamic efferents navigate correctly through the thalamus but fail to turn laterally into the telencephalon, whereas other axons are able to cross the diencephalic/telencephalic boundary. Our results support a role for the ventral telencephalon in guiding the early development of the thalamocortical tract and identify a new role for the transcription factor Pax6 in regulating the ability of the thalamus to attract ventral telencephalic afferents.
Figures

References
-
- Angevine JB., Jr Time of neuron origin in the diencephalon of the mouse. An autoradiographic study. J Comp Neurol. 1970;139:129–187. - PubMed
-
- Auladell C, Perez-Sust P, Super H, Soriano E. The early development of thalamocortical and corticothalamic projections in the mouse. Anat Embryol (Berl) 2000;201:169–179. - PubMed
-
- Braisted JE, Tuttle R, O'Leary DD. Thalamocortical axons are influenced by chemorepellent and chemoattractant activities localized to decision points along their path. Dev Biol. 1999;208:430–440. - PubMed
-
- Collinson JM, Hill RE, West JD. Different roles for Pax6 in the optic vesicle and facial epithelium mediate early morphogenesis of the murine eye. Development. 2000;127:945–956. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
