IEX-1: a new ERK substrate involved in both ERK survival activity and ERK activation
- PMID: 12356731
- PMCID: PMC129026
- DOI: 10.1093/emboj/cdf488
IEX-1: a new ERK substrate involved in both ERK survival activity and ERK activation
Abstract
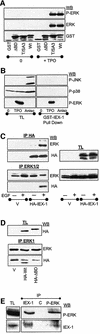
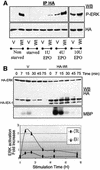
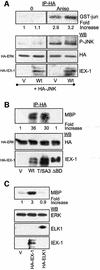
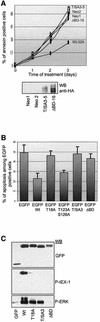
IEX-1 is an early response and NF-kappaB target gene implicated in the regulation of cellular viability. We show here that IEX-1 is a substrate for ERKs and that IEX-1 and ERK regulate each other's activities. IEX-1 was isolated by phosphorylation screening with active ERK2 and found subsequently phosphorylated in vivo upon ERK activation. IEX-1 interacts with phosphorylated ERKs but not with c-jun N-terminal kinase (JNK) or p38. Upon phosphorylation by ERKs, IEX-1 acquires the ability to inhibit cell death induced by various stimuli. In turn, IEX-1 potentiates ERK activation in response to various growth factors. By using various IEX-1 mutants in which the ERK phosphoacceptor and/or ERK docking sites were mutated, we show that the IEX-1 pro-survival effect is dependent on its phosphorylation state but not on its ability to potentiate ERK activation. Conversely, IEX-1-induced modulation of ERK activation requires ERK-IEX-1 association but is independent of IEX-1 phosphorylation. Thus, IEX-1 is a new type of ERK substrate that has a dual role in ERK signaling by acting both as an ERK downstream effector mediating survival and as a regulator of ERK activation.
Figures






Similar articles
-
Thrombopoietin regulates IEX-1 gene expression through ERK-induced AML1 phosphorylation.Blood. 2006 Apr 15;107(8):3106-13. doi: 10.1182/blood-2005-07-2953. Epub 2005 Dec 20. Blood. 2006. PMID: 16368886
-
Inhibition of B56-containing protein phosphatase 2As by the early response gene IEX-1 leads to control of Akt activity.J Biol Chem. 2007 Feb 23;282(8):5468-77. doi: 10.1074/jbc.M609712200. Epub 2007 Jan 2. J Biol Chem. 2007. PMID: 17200115
-
Compartment-specific regulation of extracellular signal-regulated kinase (ERK) and c-Jun N-terminal kinase (JNK) mitogen-activated protein kinases (MAPKs) by ERK-dependent and non-ERK-dependent inductions of MAPK phosphatase (MKP)-3 and MKP-1 in differentiating P19 cells.Biochem J. 2000 Dec 15;352 Pt 3(Pt 3):701-8. Biochem J. 2000. PMID: 11104676 Free PMC article.
-
Roles of the stress-induced gene IEX-1 in regulation of cell death and oncogenesis.Apoptosis. 2003 Jan;8(1):11-8. doi: 10.1023/a:1021688600370. Apoptosis. 2003. PMID: 12510147 Review.
-
A novel vitamin D-regulated immediate-early gene, IEX-1, alters cellular growth and apoptosis.Recent Results Cancer Res. 2003;164:123-34. doi: 10.1007/978-3-642-55580-0_8. Recent Results Cancer Res. 2003. PMID: 12899517 Free PMC article. Review.
Cited by
-
Dual PI3K/ERK inhibition induces necroptotic cell death of Hodgkin Lymphoma cells through IER3 downregulation.Sci Rep. 2016 Oct 21;6:35745. doi: 10.1038/srep35745. Sci Rep. 2016. PMID: 27767172 Free PMC article.
-
The stress-inducible immediate-early responsive gene IEX-1 is activated in cells infected with herpes simplex virus 1, but several viral mechanisms, including 3' degradation of its RNA, preclude expression of the gene.J Virol. 2003 Jun;77(11):6178-87. doi: 10.1128/jvi.77.11.6178-6187.2003. J Virol. 2003. PMID: 12743274 Free PMC article.
-
Regulation of tumor cell - Microenvironment interaction by the autotaxin-lysophosphatidic acid receptor axis.Adv Biol Regul. 2019 Jan;71:183-193. doi: 10.1016/j.jbior.2018.09.008. Epub 2018 Sep 16. Adv Biol Regul. 2019. PMID: 30243984 Free PMC article. Review.
-
G1/S cell cycle arrest provides anoikis resistance through Erk-mediated Bim suppression.Mol Cell Biol. 2005 Jun;25(12):5282-91. doi: 10.1128/MCB.25.12.5282-5291.2005. Mol Cell Biol. 2005. PMID: 15923641 Free PMC article.
-
Recent Advances in Substrate Identification of Protein Kinases in Plants and Their Role in Stress Management.Curr Genomics. 2017 Dec;18(6):523-541. doi: 10.2174/1389202918666170228142703. Curr Genomics. 2017. PMID: 29204081 Free PMC article. Review.
References
-
- Arlt A., Grobe,O., Sieke,A., Kruse,M.L., Folsch,U.R., Schmidt,W.E. and Schafer,H. (2001) Expression of the NF-κ B target gene IEX-1 (p22/PRG1) does not prevent cell death but instead triggers apoptosis in HeLa cells. Oncogene, 20, 69–76. - PubMed
-
- Ballif B.A. and Blenis,J. (2001) Molecular mechanisms mediating mammalian mitogen-activated protein kinase (MAPK) kinase (MEK)-MAPK cell survival signals. Cell Growth Differ., 12, 397–408. - PubMed
-
- Ben-Levy R., Hooper,S., Wilson,R., Paterson,H.F. and Marshall,C.J. (1998) Nuclear export of the stress-activated protein kinase p38 mediated by its substrate MAPKAP kinase-2. Curr. Biol., 8, 1049–1057. - PubMed
-
- Bonni A., Brunet,A., West,A.E., Datta,S.R., Takasu,M.A. and Greenberg,M.E. (1999) Cell survival promoted by the Ras-MAPK signaling pathway by transcription-dependent and -independent mechanisms. Science, 286, 1358–1362. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

