Decreased inducibility of TNF expression in lipid-loaded macrophages
- PMID: 12366867
- PMCID: PMC130030
- DOI: 10.1186/1471-2172-3-13
Decreased inducibility of TNF expression in lipid-loaded macrophages
Abstract
Background: Inflammation and immune responses are considered to be very important in the pathogenesis of atherosclerosis. Lipid accumulation in macrophages of the arterial intima is a characteristic feature of atherosclerosis which can influence the inflammatory potential of macrophages. We studied the effects of lipid loading on the regulation of TNF expression in human monocyte-derived macrophages.
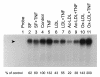
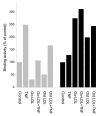
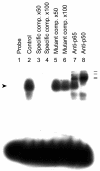
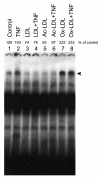
Results: In macrophages incubated with acetylated low density lipoprotein (ac-LDL) for 2 days, mRNA expression of TNF in cells stimulated with TNF decreased by 75%. In cell cultures stimulated over night with IL-1beta, lipid loading decreased secretion of TNF into culture medium by 48%. These results suggest that lipid accumulation in macrophages makes them less responsive to inflammatory stimuli. Decreased basal activity and inducibility of transcription factor AP-1 was observed in lipid-loaded cells, suggesting a mechanism for the suppression of cytokine expression. NF-kappaB binding activity and inducibility were only marginally affected by ac-LDL. LDL and ac-LDL did not activate PPARgamma. In contrast, oxidized LDL stimulated AP-1 and PPARgamma but inhibited NF-kappaB, indicating that the effects of lipid loading with ac-LDL were not due to oxidation of lipids.
Conclusions: Accumulation of lipid, mainly cholesterol, results in down-regulation of TNF expression in macrophages. Since monocytes are known to be activated by cell adhesion, these results suggest that foam cells in atherosclerotic plaques may contribute less potently to an inflammatory reaction than newly arrived monocytes/macrophages.
Figures



References
-
- Leitman DC, Ribeiro RCJ, Mackow ER, Baxter JD, West BL. Identification of a tumor necrosis factor-responsive element in the tumor necrosis factor α gene. J Biol Chem. 1991;266:9343–9346. - PubMed
-
- Jovinge S, Ares MPS, Kallin B, Nilsson J. Human monocytes/macrophages release TNF-α in response to ox-LDL. Arterioscler Thromb Vasc Biol. 1996;16:1573–1579. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

