Stable hepatitis C virus RNA detection by RT-PCR during four days storage
- PMID: 12366870
- PMCID: PMC130053
- DOI: 10.1186/1471-2334-2-22
Stable hepatitis C virus RNA detection by RT-PCR during four days storage
Abstract
Background: Suboptimal specimen processing and storage conditions of samples which contain hepatitis C virus (HCV) RNA may result in a decline of HCV RNA concentration or false-negative results in the detection of HCV RNA in serum. We evaluated the stability of HCV RNA in serum and clotted blood samples stored at room temperature or at 4 degrees C for 4 days with the aim of optimizing the standard procedures of processing and storage of samples.
Methods: Blood from five HCV RNA positive patients was collected in tubes with and without separator gel, centrifuged 1 or 6 hours after collection. Samples were then left 6, 24, 48, 72 or 96 h at room temperature (21.5 - 25.4 degrees C) or at 4 degrees C before determining their HCV RNA level using the COBAS AMPLICOR HCV MONITOR Test, vs 2.0 (Roche Diagnostic Systems).
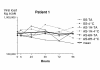
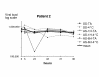
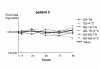
Results: The logarithm of the HCV RNA level measurements remained within a 0.3 value of the means for 4 days at both temperatures (room temperature or 4 degrees C).
Conclusions: We conclude that blood samples may be collected and aliquoted within 6 h of collection and can be stored at 4 degrees C for 72 hours as proposed by the manufacturer without significant differences in measured HCV RNA level. Our results indicate that lapses in this scheme may still yield reliable results.
Figures


Similar articles
-
Biological qualification of blood units: considerations about the effects of sample's handling and storage on stability of nucleic acids.Transfus Apher Sci. 2004 Jun;30(3):197-203. doi: 10.1016/j.transci.2003.11.010. Transfus Apher Sci. 2004. PMID: 15172624
-
[Stability of hepatitis C virus RNA in various processing and storage conditions].Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2006 Dec;14(6):1238-43. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2006. PMID: 17204202 Chinese.
-
Evaluation of hepatitis C virus RNA stability in room temperature and multiple freeze-thaw cycles by COBAS AmpliPrep/COBAS TaqMan HCV.Diagn Microbiol Infect Dis. 2013 Jan;75(1):81-5. doi: 10.1016/j.diagmicrobio.2012.09.017. Epub 2012 Oct 26. Diagn Microbiol Infect Dis. 2013. PMID: 23102559
-
Impact of various handling and storage conditions on quantitative detection of hepatitis C virus RNA.J Hepatol. 1996 Sep;25(3):307-11. doi: 10.1016/s0168-8278(96)80116-4. J Hepatol. 1996. PMID: 8895009
-
Storage conditions of blood samples and primer selection affect the yield of cDNA polymerase chain reaction products of hepatitis C virus.J Clin Microbiol. 1992 Dec;30(12):3220-4. doi: 10.1128/jcm.30.12.3220-3224.1992. J Clin Microbiol. 1992. PMID: 1333489 Free PMC article.
Cited by
-
Stability of hepatitis C virus RNA in blood samples by TaqMan real-time PCR.J Clin Lab Anal. 2010;24(3):134-8. doi: 10.1002/jcla.20354. J Clin Lab Anal. 2010. PMID: 20486191 Free PMC article.
-
Comparison of serial Hepatitis C virus detection in samples submitted through serology for reflex confirmation versus samples directly submitted for quantitation.J Clin Microbiol. 2011 Aug;49(8):3036-9. doi: 10.1128/JCM.00577-11. Epub 2011 Jun 8. J Clin Microbiol. 2011. PMID: 21653774 Free PMC article.
-
HCV RNA Testing of Plasma Samples from Cornea Donors: Suitability of Plasma Samples Stored at 4 °C for up to 8 Days.Transfus Med Hemother. 2017 Jan;44(1):39-44. doi: 10.1159/000449207. Epub 2016 Nov 4. Transfus Med Hemother. 2017. PMID: 28275332 Free PMC article.
-
Stability of hepatitis B virus pregenomic RNA in plasma specimens under various temperatures and storage conditions.PeerJ. 2021 Apr 14;9:e11207. doi: 10.7717/peerj.11207. eCollection 2021. PeerJ. 2021. PMID: 33954043 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

