Baculovirus infection raises the level of TATA-binding protein that colocalizes with viral DNA replication sites
- PMID: 12368354
- PMCID: PMC136646
- DOI: 10.1128/jvi.76.21.11123-11127.2002
Baculovirus infection raises the level of TATA-binding protein that colocalizes with viral DNA replication sites
Abstract
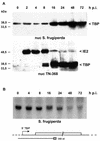
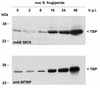
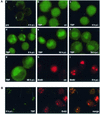
During the infection cycle of Autographa californica multicapsid nuclear polyhedrosis virus, the TATA-binding protein (TBP) of the insect host cell likely participates in early viral transcription, which is mediated by the host RNA polymerase II. However, the role of TBP in late and very late viral transcription, which is accomplished by an alpha-amanitin-resistant RNA polymerase, is unclear. We observed a dramatic increase of TBP protein during the late phases of infection. TBP mRNA levels, however, were not coordinately increased. Indirect-immunofluorescence studies revealed a nuclear redistribution of TBP during infection. After labeling of viral replication centers with bromodeoxyuridine (BrdU), costaining of TBP and BrdU showed that TBP localized to viral DNA replication centers. These results suggest a putative role of TBP during late viral transcription, which may occur in close proximity to viral DNA replication.
Figures
Similar articles
-
Expression and nuclear localization of the TATA-box-binding protein during baculovirus infection.J Gen Virol. 2014 Jun;95(Pt 6):1396-1407. doi: 10.1099/vir.0.059949-0. Epub 2014 Mar 27. J Gen Virol. 2014. PMID: 24676420
-
Nuclear IE2 structures are related to viral DNA replication sites during baculovirus infection.J Virol. 2002 May;76(10):5198-207. doi: 10.1128/jvi.76.10.5198-5207.2002. J Virol. 2002. PMID: 11967334 Free PMC article.
-
Autographa californica nucleopolyhedrovirus infection results in Sf9 cell cycle arrest at G2/M phase.Virology. 1998 Apr 25;244(1):195-211. doi: 10.1006/viro.1998.9097. Virology. 1998. PMID: 9581791
-
[TBP, a universal transcription factor?].Med Sci (Paris). 2004 May;20(5):575-9. doi: 10.1051/medsci/2004205575. Med Sci (Paris). 2004. PMID: 15190478 Review. French.
-
TBP homologues in embryo transcription: who does what?EMBO Rep. 2007 Nov;8(11):1016-8. doi: 10.1038/sj.embor.7401093. EMBO Rep. 2007. PMID: 17972900 Free PMC article. Review. No abstract available.
Cited by
-
Combined extrinsic and intrinsic manipulations exert complementary neuronal enrichment in embryonic rat neural precursor cultures: an in vitro and in vivo analysis.J Comp Neurol. 2009 Jul 1;515(1):56-71. doi: 10.1002/cne.22027. J Comp Neurol. 2009. PMID: 19399893 Free PMC article.
-
Studies of the silencing of baculovirus DNA binding protein.J Virol. 2007 Jun;81(11):6122-7. doi: 10.1128/JVI.02768-06. Epub 2007 Mar 21. J Virol. 2007. PMID: 17376916 Free PMC article.
-
Identification of Regulatory Host Genes Involved in Sigma Virus Replication Using RNAi Knockdown in Drosophila.Insects. 2019 Oct 11;10(10):339. doi: 10.3390/insects10100339. Insects. 2019. PMID: 31614679 Free PMC article.
-
Comprehensive analysis of host gene expression in Autographa californica nucleopolyhedrovirus-infected Spodoptera frugiperda cells.Virology. 2011 Mar 30;412(1):167-78. doi: 10.1016/j.virol.2011.01.006. Epub 2011 Jan 28. Virology. 2011. PMID: 21276998 Free PMC article.
-
ERK- and JNK-dependent signaling pathways contribute to Bombyx mori nucleopolyhedrovirus infection.J Virol. 2007 Dec;81(24):13700-9. doi: 10.1128/JVI.01683-07. Epub 2007 Oct 3. J Virol. 2007. PMID: 17913811 Free PMC article.
References
-
- Carstens, E. B., S. T. Tjia, and W. Doerfler. 1979. Infection of Spodoptera frugiperda cells with Autographa californica nuclear polyhedrosis virus. I. Synthesis of intracellular proteins after virus infection. Virology 99:386-398. - PubMed
-
- Harland, L., R. Crombie, S. Anson, J. deBoer, P. A. Ioannou, and M. Antoniou. 2002. Transcriptional regulation of the human TATA binding protein gene. Genomics 79:479-482. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

