Endogenous lipid- and peptide-derived anti-inflammatory pathways generated with glucocorticoid and aspirin treatment activate the lipoxin A4 receptor
- PMID: 12368905
- PMCID: PMC2777269
- DOI: 10.1038/nm786
Endogenous lipid- and peptide-derived anti-inflammatory pathways generated with glucocorticoid and aspirin treatment activate the lipoxin A4 receptor
Abstract
Aspirin (ASA) and dexamethasone (DEX) are widely used anti-inflammatory agents yet their mechanism(s) for blocking polymorphonuclear neutrophil (PMN) accumulation at sites of inflammation remains unclear. Here, we report that inhibition of PMN infiltration by ASA and DEX is a property shared by aspirin-triggered lipoxins (ATL) and the glucocorticoid-induced annexin 1 (ANXA1)-derived peptides that are both generated in vivo and act at the lipoxin A(4) receptor (ALXR/FPRL1) to halt PMN diapedesis. These structurally diverse ligands specifically interact directly with recombinant human ALXR demonstrated by specific radioligand binding and function as well as immunoprecipitation of PMN receptors. In addition, the combination of both ATL and ANXA1-derived peptides limited PMN infiltration and reduced production of inflammatory mediators (that is, prostaglandins and chemokines) in vivo. Together, these results indicate functional redundancies in endogenous lipid and peptide anti-inflammatory circuits that are spatially and temporally separate, where both ATL and specific ANXA1-derived peptides act in concert at ALXR to downregulate PMN recruitment to inflammatory loci.
Figures
 , +DEX;
, +DEX;  , +ASA; ■, +DEX & ASA.
, +ASA; ■, +DEX & ASA.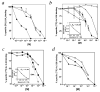
 ) or aspirin-triggered lipoxin A4 analog 1 (ATLa1; 100 nM; ■) to the lower compartment. □, vehicle. b, CHO-Gqo-ALXR cells were pretreated with Ac2-26 peptide for 30 min at 37 °C and added to the upper compartment of a microchamber (5 × 104 cells per well). Chemotaxis was initiated by addition of ATLa1 (100 nM) to the lower compartment. c, Human PMNs were added to the upper compartment of a microchamber (5 × 104 cells per well). Chemotaxis was initiated by addition of Ac2-26 peptide (1–10 μM; ■) to the lower compartment. In some cases, cells were treated with 10 (
) or aspirin-triggered lipoxin A4 analog 1 (ATLa1; 100 nM; ■) to the lower compartment. □, vehicle. b, CHO-Gqo-ALXR cells were pretreated with Ac2-26 peptide for 30 min at 37 °C and added to the upper compartment of a microchamber (5 × 104 cells per well). Chemotaxis was initiated by addition of ATLa1 (100 nM) to the lower compartment. c, Human PMNs were added to the upper compartment of a microchamber (5 × 104 cells per well). Chemotaxis was initiated by addition of Ac2-26 peptide (1–10 μM; ■) to the lower compartment. In some cases, cells were treated with 10 ( ) or 100 nM (□) ATLa1 for 30 min at 37 °C. #, P = 0.01, versus vehicle; *, P < 0.01, versus Ac2-26 alone (a and c) or % inhibition of ATLa1- evoked chemotaxis (*, P = 0.02; **, P < 0.01, versus ATLa1 alone in b). Data represent the mean ± s.e.m. from n = 3 experiments. d, Fura2-AM-loaded human PMNs (5 × 106 cells/incubation) were incubated with the different stimuli, and rapid changes in intracellular [Ca2+] measured by fluorimetry. Additions of agonists are denoted by arrows. hrANXA1: human recombinant annexin 1. Traces are representative of 3 independent experiments.
) or 100 nM (□) ATLa1 for 30 min at 37 °C. #, P = 0.01, versus vehicle; *, P < 0.01, versus Ac2-26 alone (a and c) or % inhibition of ATLa1- evoked chemotaxis (*, P = 0.02; **, P < 0.01, versus ATLa1 alone in b). Data represent the mean ± s.e.m. from n = 3 experiments. d, Fura2-AM-loaded human PMNs (5 × 106 cells/incubation) were incubated with the different stimuli, and rapid changes in intracellular [Ca2+] measured by fluorimetry. Additions of agonists are denoted by arrows. hrANXA1: human recombinant annexin 1. Traces are representative of 3 independent experiments.
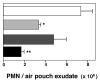
 , + 3 μg DEX;
, + 3 μg DEX;  , + 300 μg ASA; ■, + 3 μg DEX + 300 μg ASA.
, + 300 μg ASA; ■, + 3 μg DEX + 300 μg ASA.
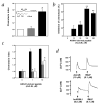
 ) 15 min before local injection of Zymosan A. PMN accumulation was measured after 4 h. The number of migrated PMNs in the control group (mice treated with Zymosan A alone) was 6.5 ± 0.55 × 106 PMNs per mouse. Data are mean ± s.e.m of n = 6–8 mice per group; *, P < 0.05 versus control migration as calculated on original values. c, Air-pouch biopsies. Sections from the top dose groups (that is, 5 μg of ATLa and 100 μg of Ac2-26; see b) were prepared and were stained with H&E. Magnifications, ×10 for the top panel and ×40 for the inset and lower panels.
) 15 min before local injection of Zymosan A. PMN accumulation was measured after 4 h. The number of migrated PMNs in the control group (mice treated with Zymosan A alone) was 6.5 ± 0.55 × 106 PMNs per mouse. Data are mean ± s.e.m of n = 6–8 mice per group; *, P < 0.05 versus control migration as calculated on original values. c, Air-pouch biopsies. Sections from the top dose groups (that is, 5 μg of ATLa and 100 μg of Ac2-26; see b) were prepared and were stained with H&E. Magnifications, ×10 for the top panel and ×40 for the inset and lower panels.References
-
- Cotran RS. Inflammation: Historical perspectives. In: Gallin JI, Snyderman R, Fearon DT, Haynes BF, Nathan C, editors. Inflammation: Basic Principles and Clinical Correlates. 3rd edn Lippincott Williams & Wilkins; Philadelphia: 1999. pp. 5–10.
-
- Weissmann G. Aspirin. Sci. Am. 1991;264:84–90. - PubMed
-
- Patrono C. Aspirin: new cardiovascular uses for an old drug. Am. J. Med. 2001;110:62S–65S. - PubMed
-
- Barnes CJ, Hardman WE, Cameron IL, Lee M. Aspirin, but not sodium salicylate, indomethacin, or nabumetone, reversibly suppresses 1,2-dimethylhydrazine-induced colonic aberrant crypt foci in rats. Dig. Dis. Sci. 1997;42:920–926. - PubMed
-
- Marcus AJ. Aspirin as prophylaxis against colorectal cancer. N. Engl. J. Med. 1995;333:656–658. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

