Structure and function in rhodopsin: a tetracycline-inducible system in stable mammalian cell lines for high-level expression of opsin mutants
- PMID: 12370422
- PMCID: PMC129687
- DOI: 10.1073/pnas.212519199
Structure and function in rhodopsin: a tetracycline-inducible system in stable mammalian cell lines for high-level expression of opsin mutants
Abstract
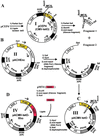
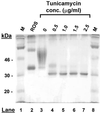
Tetracycline-inducible HEK293S stable cell lines have been prepared that express high levels (up to 10 mg/liter) of WT opsin and its mutants only in response to the addition of tetracycline and sodium butyrate. The cell lines were prepared by stable transfection of HEK293S-TetR cells with expression plasmids that contained the opsin gene downstream of a cytomegalovirus promoter containing tetO sequences as well as the neomycin resistance gene under control of the weak H(2)L(d) promoter. The inducible system is particularly suited for overcoming problems with toxicity either due to the addition of toxic compounds, for example, tunicamycin, to the growth medium or due to the expressed protein products. By optimization of cell growth conditions in a bioreactor, WT opsin, a constitutively active opsin mutant, E113Q/E134Q/M257Y, presumed to be toxic to the cells, and nonglycosylated WT opsin obtained by growth in the presence of tunicamycin have been prepared in amounts of several milligrams per liter of culture medium.
Figures




Similar articles
-
Structure and function in rhodopsin: high-level expression of rhodopsin with restricted and homogeneous N-glycosylation by a tetracycline-inducible N-acetylglucosaminyltransferase I-negative HEK293S stable mammalian cell line.Proc Natl Acad Sci U S A. 2002 Oct 15;99(21):13419-24. doi: 10.1073/pnas.212519299. Epub 2002 Oct 7. Proc Natl Acad Sci U S A. 2002. PMID: 12370423 Free PMC article.
-
Structure and function in rhodopsin: high level expression of a synthetic bovine opsin gene and its mutants in stable mammalian cell lines.Proc Natl Acad Sci U S A. 1996 Oct 15;93(21):11487-92. doi: 10.1073/pnas.93.21.11487. Proc Natl Acad Sci U S A. 1996. PMID: 8876162 Free PMC article.
-
Development of stable cell lines expressing high levels of point mutants of human opsin for biochemical and biophysical studies.Methods Enzymol. 2000;315:30-58. doi: 10.1016/s0076-6879(00)15833-1. Methods Enzymol. 2000. PMID: 10736692
-
Opsin genes.Essays Biochem. 1995;29:87-111. Essays Biochem. 1995. PMID: 9189715 Review. No abstract available.
-
Expression systems for bovine rhodopsin: a review of the progress made in the Khorana laboratory.Biophys Rev. 2023 Jan 6;15(1):93-101. doi: 10.1007/s12551-022-01037-2. eCollection 2023 Feb. Biophys Rev. 2023. PMID: 36909956 Free PMC article. Review.
Cited by
-
High-level expression and purification of Cys-loop ligand-gated ion channels in a tetracycline-inducible stable mammalian cell line: GABAA and serotonin receptors.Protein Sci. 2010 Sep;19(9):1728-38. doi: 10.1002/pro.456. Protein Sci. 2010. PMID: 20662008 Free PMC article.
-
New insights into the molecular mechanism of rhodopsin retinitis pigmentosa from the biochemical and functional characterization of G90V, Y102H and I307N mutations.Cell Mol Life Sci. 2022 Jan 7;79(1):58. doi: 10.1007/s00018-021-04086-0. Cell Mol Life Sci. 2022. PMID: 34997336 Free PMC article.
-
A coiled-coil-based design strategy for the thermostabilization of G-protein-coupled receptors.Sci Rep. 2023 Jun 22;13(1):10159. doi: 10.1038/s41598-023-36855-1. Sci Rep. 2023. PMID: 37349348 Free PMC article.
-
Expression of recombinant glycoproteins in mammalian cells: towards an integrative approach to structural biology.Curr Opin Struct Biol. 2013 Jun;23(3):345-56. doi: 10.1016/j.sbi.2013.04.003. Epub 2013 Apr 25. Curr Opin Struct Biol. 2013. PMID: 23623336 Free PMC article. Review.
-
MiRNA mimic screen for improved expression of functional neurotensin receptor from HEK293 cells.Biotechnol Bioeng. 2015 Aug;112(8):1632-43. doi: 10.1002/bit.25567. Epub 2015 Jun 16. Biotechnol Bioeng. 2015. PMID: 25676429 Free PMC article.
References
-
- Reeves P J, Klein-Seetharaman J, Getmanova E V, Eilers M, Loewen M C, Smith S O, Khorana H G. Biochem Soc Trans. 1999;27:950–955. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

