The abundant class III chitinase homolog in young developing banana fruits behaves as a transient vegetative storage protein and most probably serves as an important supply of amino acids for the synthesis of ripening-associated proteins
- PMID: 12376669
- PMCID: PMC166631
- DOI: 10.1104/pp.006551
The abundant class III chitinase homolog in young developing banana fruits behaves as a transient vegetative storage protein and most probably serves as an important supply of amino acids for the synthesis of ripening-associated proteins
Abstract
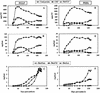
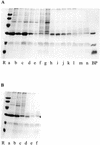
Analyses of the protein content and composition revealed dramatic changes in gene expression during in situ banana (Musa spp.) fruit formation/ripening. The total banana protein content rapidly increases during the first 60 to 70 d, but remains constant for the rest of fruit formation/ripening. During the phase of rapid protein accumulation, an inactive homolog of class III chitinases accounts for up to 40% (w/v) of the total protein. Concomitant with the arrest of net protein accumulation, the chitinase-related protein (CRP) progressively decreases and several novel proteins appear in the electropherograms. Hence, CRP behaves as a fruit-specific vegetative storage protein that accumulates during early fruit formation and serves as a source of amino acids for the synthesis of ripening-associated proteins. Analyses of individual proteins revealed that a thaumatin-like protein, a beta-1,3-glucanase, a class I chitinase, and a mannose-binding lectin are the most abundant ripening-associated proteins. Because during the ripening of prematurely harvested bananas, similar changes take place as in the in situ ripening bananas, CRP present in immature fruits is a sufficient source of amino acids for a quasi-normal synthesis of ripening-associated proteins. However, it is evident that the conversion of CRP in ripening-associated proteins takes place at an accelerated rate, especially when climacteric ripening is induced by ethylene. The present report also includes a discussion of the accumulation of the major banana allergens and the identification of suitable promoters for the production of vaccines in transgenic bananas.
Figures
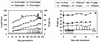


References
-
- Barre A, Peumans WJ, Menu-Bouaouiche L, Van Damme EJM, May GD, Fernandez Herrera A, Van Leuven F, Rougé P. Purification and structural analysis of an abundant thaumatin-like protein from ripe banana fruit. Planta. 2000;211:791–799. - PubMed
-
- Blanco C, Diaz-Perales A, Collada C, Sanchez-Monge R, Aragoncillo C, Castillo R, Ortega N, Alvarez M, Carrillo T, Salcedo G. Class I chitinases as potential panallergens involved in the latex-fruit syndrome. J Allergy Clin Immunol. 1999;103:507–513. - PubMed
-
- Bleecker AB, Kende H. Ethylene: a gaseous signal molecule in plants. Annu Rev Cell Dev Biol. 2000;16:1–18. - PubMed
-
- Breiteneder H, Ebner C. Molecular and biochemical classification of plant-derived food allergens. J Allergy Clin Immunol. 2000;106:27–36. - PubMed
-
- Breiteneder H, Scheiner O. Molecular and immunological characteristics of latex allergens. Int Arch Allergy Immunol. 1998;116:83–92. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

