Acquisition of regulators of complement activation by Streptococcus pyogenes serotype M1
- PMID: 12379699
- PMCID: PMC130388
- DOI: 10.1128/IAI.70.11.6206-6214.2002
Acquisition of regulators of complement activation by Streptococcus pyogenes serotype M1
Abstract
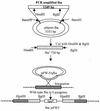
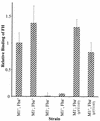
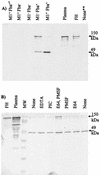
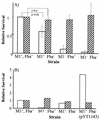
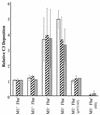
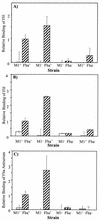
Opsonization of bacteria by complement proteins is an important component of the immune response. The pathogenic bacterium Streptococcus pyogenes has evolved multiple mechanisms for the evasion of complement-mediated opsonization. One mechanism involves the binding of human regulators of complement activation such as factor H (FH) and FH-like protein 1 (FHL-1). Acquisition of these regulatory proteins can limit deposition of the opsonin C3b on bacteria, thus decreasing the pathogen's susceptibility to phagocytosis. Binding of complement regulatory proteins by S. pyogenes has previously been attributed to the streptococcal M and M-like proteins. Here, we report that the S. pyogenes cell surface protein Fba can mediate binding of FH and FHL-1. We constructed mutant derivatives of S. pyogenes that lack Fba, M1 protein, or both proteins and assayed the strains for FH binding, susceptibility to phagocytosis, and C3 deposition. Fba expression was found to be sufficient for binding of purified FH as well as for binding of FH and FHL-1 from human plasma. Plasma adsorption experiments also revealed that M1(+) Fba(+) streptococci preferentially bind FHL-1, whereas M1(-) Fba(+) streptococci have similar affinities for FH and FHL-1. Fba was found to contribute to the survival of streptococci incubated with human blood and to inhibit C3 deposition on bacterial cells. Streptococci harvested from log-phase cultures readily bound FH, but binding was greatly reduced for bacteria obtained from stationary-phase cultures. Bacteria cultured in the presence of the protease inhibitor E64 maintained FH binding activity in stationary phase, suggesting that Fba is removed from the cell surface via proteolysis. Western analyses confirmed that E64 stabilizes cell surface expression of Fba. These data indicate that Fba is an antiopsonic, antiphagocytic protein that may be regulated by cell surface proteolysis.
Figures

References
-
- Berge, A., and L. Bjorck. 1995. Streptococcal cysteine proteinase releases biologically active fragments of streptococcal surface proteins. J. Biol. Chem. 270:9862-9867. - PubMed
-
- Berge, A., B. M. Kihlberg, A. G. Sjoholm, and L. Bjorck. 1997. Streptococcal protein H forms soluble complement-activating complexes with IgG, but inhibits complement activation by IgG-coated targets. J. Biol. Chem. 272:20774-20781. - PubMed
-
- Caparon, M. G., and J. R. Scott. 1991. Genetic manipulation of pathogenic streptococci. Methods Enzymol. 204:556-586. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

