Immunization with C5a peptidase or peptidase-type III polysaccharide conjugate vaccines enhances clearance of group B Streptococci from lungs of infected mice
- PMID: 12379721
- PMCID: PMC130386
- DOI: 10.1128/IAI.70.11.6409-6415.2002
Immunization with C5a peptidase or peptidase-type III polysaccharide conjugate vaccines enhances clearance of group B Streptococci from lungs of infected mice
Abstract
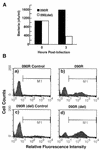
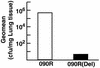
Group B streptococci (GBS) are among the most common causes of life-threatening neonatal infections. Vaccine development since the late 1970s has focused on the capsular polysaccharides, but a safe, effective product is still not available. Our quest for a vaccine turned to the streptococcal C5a peptidase (SCPB). This surface protein is antigenically conserved across most if not all serotypes. A murine model was used to assess the impact of SCPB on clearance of GBS from the lungs of intranasally infected animals. Mutational inactivation of SCPB resulted in more-rapid clearance of streptococci from the lung. Immunization with recombinant SCPB alone or SCPB conjugated to type III capsular polysaccharide produced serotype-independent protection, which was evidenced by more-rapid clearance of the serotype VI strain from the lungs. Immunization of mice with tetanus toxoid-type III polysaccharide conjugate did not produce protection, confirming that protection induced by SCPB conjugates was independent of type III polysaccharide antigen. Histological evaluation of lungs from infected mice revealed that pathology in animals immunized with SCPB or SCPB conjugates was significantly less than that in animals immunized with a tetanus toxoid-polysaccharide conjugate. These experiments suggest that inclusion of C5a peptidase in a vaccine will both add another level to and broaden the spectrum of the protection of a polysaccharide vaccine.
Figures



References
-
- Ablow, R. C., S. G. Driscoll, E. L. Effmann, I. Gross, C. J. Jolles, R. Uauy, and J. B. Warshaw. 1976. A comparison of early-onset group B streptococcal neonatal infection and the respiratory-distress syndrome of the newborn. N. Engl. J. Med. 294:65-70. - PubMed
-
- Baker, C. J., and M. S. Edwards. 1995. Group B streptococcal infections, p. 980-1054. In J. S. Remington and J. O. Klein (ed.), Infectious diseases of the fetus and newborn infant, 4th ed. W. B. Saunders Co., Philadelphia, Pa.
-
- Baker, C. J., N. A. Halsey, and A. Schuchat. 1999. 1997 AAP guidelines for prevention of early-onset group B streptococcal disease. Pediatrics 103:197-198. - PubMed
-
- Baker, C. J., and D. L. Kasper. 1976. Correlation of maternal antibody deficiency with susceptibility to neonatal group B streptococcal infection. N. Engl. J. Med. 294:753-756. - PubMed
-
- Baker, C. J., and D. L. Kasper. 1985. Group B streptococcal vaccines. Rev. Infect. Dis. 7:458-467. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

