Development and evolution of inner ear sensory epithelia and their innervation
- PMID: 12382272
- PMCID: PMC4943216
- DOI: 10.1002/neu.10098
Development and evolution of inner ear sensory epithelia and their innervation
Abstract
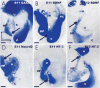
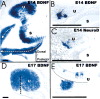
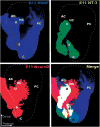
The development and evolution of the inner ear sensory patches and their innervation is reviewed. Recent molecular developmental data suggest that development of these sensory patches is a developmental recapitulation of the evolutionary history. These data suggest that the ear generates multiple, functionally diverse sensory epithelia by dividing a single sensory primordium. Those epithelia will establish distinct identities through the overlapping expression of genes of which only a few are currently known. One of these distinctions is the unique pattern of hair cell polarity. A hypothesis is presented on how the hair cell polarity may relate to the progressive segregation of the six sensory epithelia. Besides being markers for sensory epithelia development, neurotrophins are also expressed in delaminating cells that migrate toward the developing vestibular and cochlear ganglia. These delaminating cells originate from multiple sites at or near the developing sensory epithelia and some also express neuronal markers such as NeuroD. The differential origin of precursors raises the possibility that some sensory neurons acquire positional information before they delaminate the ear. Such an identity of these delaminating sensory neurons may be used both to navigate their dendrites to the area they delaminated from, as well as to help them navigate to their central target. The navigational properties of sensory neurons as well as the acquisition of discrete sensory patch phenotypes implies a much more sophisticated subdivision of the developing otocyst than the few available gene expression studies suggest.
Copyright 2002 Wiley Periodicals, Inc.
Figures


References
-
- Acampora D, Gulisano M, Simeone A. Genetic and molecular roles of Otx homeodomain proteins in head development. Gene. 2000;246:23–35. - PubMed
-
- Begbie J, Graham A. Integration between the epibranchial placodes and the hindbrain. Science. 2001;294:595– 598. - PubMed
-
- Bermingham NA, Hassan BA, Price SD, Vollrath MA, Ben-Arie N, Eatock RA, Bellen HJ, Lysakowski A, Zoghbi HY. Math1: an essential gene for the generation of inner ear hair cells. Science. 1999;284:1837–1841. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

