Melanization of Cryptococcus neoformans and Histoplasma capsulatum reduces their susceptibilities to amphotericin B and caspofungin
- PMID: 12384341
- PMCID: PMC128748
- DOI: 10.1128/AAC.46.11.3394-3400.2002
Melanization of Cryptococcus neoformans and Histoplasma capsulatum reduces their susceptibilities to amphotericin B and caspofungin
Abstract
The fungal pathogens Cryptococcus neoformans and Histoplasma capsulatum produce melanin-like pigments in the presence of L-dopa in vitro and during mammalian infection. We investigated whether melanization affected the susceptibilities of the fungi to amphotericin B, caspofungin, fluconazole, itraconazole, or flucytosine (5FC). Using the standard macrodilution MIC protocol (the M27A protocol) of the National Committee for Clinical Laboratory Standards for yeast, we found no difference in the susceptibilities of melanized and nonmelanized C. neoformans and H. capsulatum isolates. Killing assays demonstrated that melanization reduced the susceptibilities of both fungi to amphotericin B and caspofungin. Laccase-deficient C. neoformans cells grown with L-dopa were significantly more susceptible than congenic melanin-producing yeast to killing by amphotericin B or caspofungin. Preincubation of amphotericin B or caspofungin with melanins decreased their antifungal activities. Elemental analysis of melanins incubated with amphotericin B or caspofungin revealed an alteration in the C:N ratios of the melanins, which indicated binding of these drugs by the melanins. In contrast, incubation of fluconazole, itraconazole, or 5FC with melanins did not significantly affect the antifungal efficacies of the drugs or the chemical composition of the melanins. The results suggest a potential explanation for the inefficacy of caspofungin against C. neoformans in vivo, despite activity in vitro. Furthermore, the results indicate that fungal melanins protect C. neoformans and H. capsulatum from the activities of amphotericin B and caspofungin and that this protection is not demonstrable by standard broth macrodilution assays.
Figures
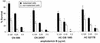
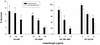
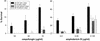

Similar articles
-
Susceptibility of Cryptococcus neoformans biofilms to antifungal agents in vitro.Antimicrob Agents Chemother. 2006 Mar;50(3):1021-33. doi: 10.1128/AAC.50.3.1021-1033.2006. Antimicrob Agents Chemother. 2006. PMID: 16495265 Free PMC article.
-
Cryptococcus neoformans/Cryptococcus gattii species complex melanized by epinephrine: Increased yeast survival after amphotericin B exposure.Microb Pathog. 2020 Jun;143:104123. doi: 10.1016/j.micpath.2020.104123. Epub 2020 Mar 10. Microb Pathog. 2020. PMID: 32169493
-
Pneumocandin L-743,872 enhances the activities of amphotericin B and fluconazole against Cryptococcus neoformans in vitro.Antimicrob Agents Chemother. 1997 Feb;41(2):331-6. doi: 10.1128/AAC.41.2.331. Antimicrob Agents Chemother. 1997. PMID: 9021188 Free PMC article.
-
Melanin and fungi.Curr Opin Infect Dis. 2003 Apr;16(2):91-6. doi: 10.1097/00001432-200304000-00005. Curr Opin Infect Dis. 2003. PMID: 12734441 Review.
-
Caspofungin: first approved agent in a new class of antifungals.Expert Opin Pharmacother. 2003 May;4(5):807-23. doi: 10.1517/14656566.4.5.807. Expert Opin Pharmacother. 2003. PMID: 12740003 Review.
Cited by
-
Virulence Factors as Targets for Anticryptococcal Therapy.J Fungi (Basel). 2016 Nov 30;2(4):29. doi: 10.3390/jof2040029. J Fungi (Basel). 2016. PMID: 29376946 Free PMC article. Review.
-
Antifungal therapeutics for dimorphic fungal pathogens.Virulence. 2017 Feb 17;8(2):211-221. doi: 10.1080/21505594.2016.1235653. Epub 2016 Sep 19. Virulence. 2017. PMID: 27646561 Free PMC article.
-
Transposable element insertions shape gene regulation and melanin production in a fungal pathogen of wheat.BMC Biol. 2018 Jul 16;16(1):78. doi: 10.1186/s12915-018-0543-2. BMC Biol. 2018. PMID: 30012138 Free PMC article.
-
Cryptococcus neoformans resistance to echinocandins: (1,3)beta-glucan synthase activity is sensitive to echinocandins.Antimicrob Agents Chemother. 2005 Jul;49(7):2851-6. doi: 10.1128/AAC.49.7.2851-2856.2005. Antimicrob Agents Chemother. 2005. PMID: 15980360 Free PMC article.
-
Deciphering the Role of PIG1 and DHN-Melanin in Scedosporium apiospermum Conidia.J Fungi (Basel). 2023 Jan 18;9(2):134. doi: 10.3390/jof9020134. J Fungi (Basel). 2023. PMID: 36836250 Free PMC article.
References
-
- Abruzzo, G. K., A. M. Flattery, C. J. Gill, L. Kong, J. G. Smith, V. B. Pikounis, J. M. Balkovec, A. F. Bouffard, J. F. Dropinski, H. Rosen, H. Kropp, and K. Bartizal. 1997. Evaluation of the echinocandin antifungal MK-0991 (L-743,872): efficacies in mouse models of disseminated aspergillosis, candidiasis, and cryptococcosis. Antimicrob. Agents Chemother. 41:2333-2338. - PMC - PubMed
-
- Avramidis, N., A. Kourounakis, L. Hadjipetrou, and V. Senchuk. 1998. Anti-inflammatory and immunomodulating properties of grape melanin. Inhibitory effects on paw edema and adjuvant induced disease. Arzneimittelforschung 48:764-771. - PubMed
-
- Bouffard, F. A., R. A. Zambias, J. F. Dropinski, J. M. Balkovec, M. L. Hammond, G. K. Abruzzo, K. F. Bartizal, J. A. Marrinan, M. B. Kurtz, D. C. McFadden, et al. 1994. Synthesis and antifungal activity of novel cationic pneumocandin B(o) derivatives. J. Med. Chem. 37:222-225. - PubMed
-
- Casadevall, A., A. L. Rosas, and J. D. Nosanchuk. 2000. Melanin and virulence in Cryptococcus neoformans. Curr. Opin. Microbiol. 3:354-358. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

