Proinflammatory and cytotoxic effects of hexadecylphosphocholine (miltefosine) against drug-resistant strains of Trypanosoma cruzi
- PMID: 12384352
- PMCID: PMC128733
- DOI: 10.1128/AAC.46.11.3472-3477.2002
Proinflammatory and cytotoxic effects of hexadecylphosphocholine (miltefosine) against drug-resistant strains of Trypanosoma cruzi
Abstract
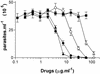
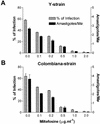
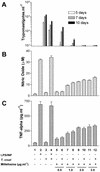
The increased resistance of the protozoan parasite Trypanosoma cruzi to nitro derivatives is one of the major problems for the successful treatment of Chagas' disease. In the present study, we have tested the effects of 1-O-hexadecylphosphocholine (miltefosine) against strains of T. cruzi that are partially resistant (strain Y) and highly resistant (strain Colombiana) to the drugs in clinical use. As expected, epimastigotes of strain Colombiana showed higher levels of resistance to benznidazole than those of strain Y. However, the level of resistance to miltefosine was the same for both strains. This alkylphospholipid was also extremely toxic against intracellular amastigotes of both strains. This ether-lipid analogue induced in a dose-dependent manner the production of tumor necrosis factor alpha and nitric oxide (NO) radicals by infected and noninfected macrophages, suggesting that miltefosine may activate macrophages in vitro. Nevertheless, the cytotoxic effect of miltefosine against intracellular amastigotes was independent of the amount of NO produced by the infected macrophages since the same dose-response curves for miltefosine were observed when the NO production was blocked by the NO synthase inhibitor N(G)-monomethyl-L-arginine monoacetate. Preliminary in vivo studies with BALB/c mice infected with strain Y indicated that oral miltefosine promoted survival and reduced the parasitemia to levels comparable to those observed when benznidazole was used. Four months after treatment, no parasites were detected in the blood or spleen tissue sections maintained in culture. Together, these results support the hypothesis that miltefosine may be used for the treatment of Chagas' disease, including cases caused by resistant strains of T. cruzi.
Figures
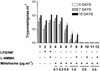

Similar articles
-
Synthesis and Evaluation of a Hybrid Miltefosine-Silver Nanoparticle Complex: Synergistic Interaction with Benznidazole Against Trypanosoma cruzi.Acta Parasitol. 2025 Jun 12;70(3):135. doi: 10.1007/s11686-025-01074-3. Acta Parasitol. 2025. PMID: 40504446 Free PMC article.
-
In vitro susceptibility of Trypanosoma cruzi strains from Santander, Colombia, to hexadecylphosphocholine (miltefosine), nifurtimox and benznidazole.Biomedica. 2009 Sep;29(3):448-55. Biomedica. 2009. PMID: 20436996
-
Activities of the triazole derivative SCH 56592 (posaconazole) against drug-resistant strains of the protozoan parasite Trypanosoma (Schizotrypanum) cruzi in immunocompetent and immunosuppressed murine hosts.Antimicrob Agents Chemother. 2000 Jan;44(1):150-5. doi: 10.1128/AAC.44.1.150-155.2000. Antimicrob Agents Chemother. 2000. PMID: 10602737 Free PMC article.
-
Targeting calcium homeostasis as the therapy of Chagas' disease and leishmaniasis - a review.Trop Biomed. 2011 Dec;28(3):471-81. Trop Biomed. 2011. PMID: 22433874 Review.
-
Unmasking the Mechanism behind Miltefosine: Revealing the Disruption of Intracellular Ca2+ Homeostasis as a Rational Therapeutic Target in Leishmaniasis and Chagas Disease.Biomolecules. 2024 Mar 27;14(4):406. doi: 10.3390/biom14040406. Biomolecules. 2024. PMID: 38672424 Free PMC article. Review.
Cited by
-
Possible mechanism of miltefosine-mediated death of Leishmania donovani.Antimicrob Agents Chemother. 2004 Aug;48(8):3010-5. doi: 10.1128/AAC.48.8.3010-3015.2004. Antimicrob Agents Chemother. 2004. PMID: 15273114 Free PMC article.
-
Experimental and Clinical Treatment of Chagas Disease: A Review.Am J Trop Med Hyg. 2017 Nov;97(5):1289-1303. doi: 10.4269/ajtmh.16-0761. Epub 2017 Oct 10. Am J Trop Med Hyg. 2017. PMID: 29016289 Free PMC article. Review.
-
Bioactivity of miltefosine against aquatic stages of Schistosoma mansoni, Schistosoma haematobium and their snail hosts, supported by scanning electron microscopy.Parasit Vectors. 2011 May 11;4:73. doi: 10.1186/1756-3305-4-73. Parasit Vectors. 2011. PMID: 21569375 Free PMC article.
-
Synthesis and Evaluation of a Hybrid Miltefosine-Silver Nanoparticle Complex: Synergistic Interaction with Benznidazole Against Trypanosoma cruzi.Acta Parasitol. 2025 Jun 12;70(3):135. doi: 10.1007/s11686-025-01074-3. Acta Parasitol. 2025. PMID: 40504446 Free PMC article.
-
A functional genomic screen in Saccharomyces cerevisiae reveals divergent mechanisms of resistance to different alkylphosphocholine chemotherapeutic agents.G3 (Bethesda). 2021 Sep 27;11(10):jkab233. doi: 10.1093/g3journal/jkab233. G3 (Bethesda). 2021. PMID: 34568930 Free PMC article.
References
-
- Andrews, N. W., M. J. M. Alves, R. I. Schumacher, and W. Colli. 1985. Trypanosoma cruzi: protection in mice immunized with 8-methoxypsolaren-inactivated trypomastigotes. Exp. Parasitol. 60:255-262. - PubMed
-
- Arana, B., N. Rizzo, and A. Diaz. 2001. Chemotherapy of cutaneous leishmaniasis: a review. Med. Microbiol. Immunol. 190:93-95. - PubMed
-
- Contreras, V. T., J. M. Salles, N. Thomas, C. M. Morel, and S. Goldenberg. 1985. In vitro differentiation of Trypanosoma cruzi under chemically defined conditions. Mol. Biochem. Parasitol. 16:315-327. - PubMed
-
- Croft, S. L., R. A. Neal, W. Pendergast, and J. H. Chan. 1987. The activity of alkyl phosphorylcholines and related derivatives against Leishmania donovani. Biochem. Pharmacol. 36:2633-2636. - PubMed
-
- Croft, S. L., D. Snowdon, and V. Yardley. 1996. The activities of four anticancer alkyllysophospholipids against Leishmania donovani, Trypanosoma cruzi and Trypanosoma brucei. J. Antimicrob. Chemother. 38:1041-1047. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

