Ca2+-controlled competitive diacylglycerol binding of protein kinase C isoenzymes in living cells
- PMID: 12391024
- PMCID: PMC2173038
- DOI: 10.1083/jcb.200203048
Ca2+-controlled competitive diacylglycerol binding of protein kinase C isoenzymes in living cells
Abstract
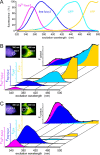
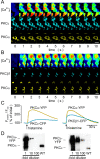
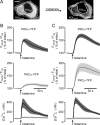
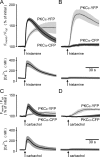
The cellular decoding of receptor-induced signaling is based in part on the spatiotemporal activation pattern of PKC isoforms. Because classical and novel PKC isoforms contain diacylglycerol (DAG)-binding C1 domains, they may compete for DAG binding. We reasoned that a Ca2+-induced membrane association of classical PKCs may accelerate the DAG binding and thereby prevent translocation of novel PKCs. Simultaneous imaging of fluorescent PKC fusion proteins revealed that during receptor stimulation, PKC alpha accumulated in the plasma membrane with a diffusion-limited kinetic, whereas translocation of PKC epsilon was delayed and attenuated. In BAPTA-loaded cells, however, a selective translocation of PKC epsilon, but not of coexpressed PKC alpha, was evident. A membrane-permeable DAG analogue displayed a higher binding affinity for PKC epsilon than for PKC alpha. Subsequent photolysis of caged Ca2+ immediately recruited PKC alpha to the membrane, and DAG-bound PKC epsilon was displaced. At low expression levels of PKC epsilon, PKC alpha concentration dependently prevented the PKC epsilon translocation with half-maximal effects at equimolar coexpression. Furthermore, translocation of endogenous PKCs in vascular smooth muscle cells corroborated the model that a competition between PKC isoforms for DAG binding occurs at native expression levels. We conclude that Ca2+-controlled competitive DAG binding contributes to the selective recruitment of PKC isoforms after receptor activation.
Figures
Similar articles
-
The catalytic domain limits the translocation of protein kinase C alpha in response to increases in Ca2+ and diacylglycerol.Biochem J. 2003 Mar 15;370(Pt 3):901-12. doi: 10.1042/BJ20021420. Biochem J. 2003. PMID: 12460119 Free PMC article.
-
Differential translocation of protein kinase C isozymes by thrombin and platelet-derived growth factor. A possible function for phosphatidylcholine-derived diacylglycerol.J Biol Chem. 1993 May 15;268(14):10534-9. J Biol Chem. 1993. PMID: 8486706
-
Protein kinase C activation during Ca2+-independent vascular smooth muscle contraction.J Surg Res. 1998 Jul 15;78(1):48-53. doi: 10.1006/jsre.1998.5368. J Surg Res. 1998. PMID: 9733617
-
Protein kinase C activation: isozyme-specific effects on metabolism and cardiovascular complications in diabetes.Diabetologia. 2001 Jun;44(6):659-73. doi: 10.1007/s001250051675. Diabetologia. 2001. PMID: 11440359 Review.
-
Subspecies-specific targeting mechanism of protein kinase C.Jpn J Pharmacol. 1998 Dec;78(4):411-7. doi: 10.1254/jjp.78.411. Jpn J Pharmacol. 1998. PMID: 9920197 Review.
Cited by
-
TRPA1 is functionally expressed in melanoma cells but is not critical for impaired proliferation caused by allyl isothiocyanate or cinnamaldehyde.Naunyn Schmiedebergs Arch Pharmacol. 2012 Jun;385(6):555-63. doi: 10.1007/s00210-012-0747-x. Epub 2012 Mar 18. Naunyn Schmiedebergs Arch Pharmacol. 2012. PMID: 22426997
-
A spatiotemporally coordinated cascade of protein kinase C activation controls isoform-selective translocation.Mol Cell Biol. 2006 Mar;26(6):2247-61. doi: 10.1128/MCB.26.6.2247-2261.2006. Mol Cell Biol. 2006. PMID: 16508001 Free PMC article.
-
Enhancing titers of therapeutic lentiviral vectors using PKC agonists.Mol Ther Methods Clin Dev. 2025 May 7;33(2):101484. doi: 10.1016/j.omtm.2025.101484. eCollection 2025 Jun 12. Mol Ther Methods Clin Dev. 2025. PMID: 40491664 Free PMC article.
-
Inhibitory modulation of distal C-terminal on protein kinase C-dependent phospho-regulation of rat TRPV1 receptors.J Physiol. 2004 Nov 1;560(Pt 3):627-38. doi: 10.1113/jphysiol.2004.069054. Epub 2004 Sep 16. J Physiol. 2004. PMID: 15375192 Free PMC article.
-
Protein kinase C isozyme-specific potentiation of expressed Ca v 2.3 currents by acetyl-beta-methylcholine and phorbol-12-myristate, 13-acetate.Brain Res. 2008 May 19;1210:1-10. doi: 10.1016/j.brainres.2008.03.017. Epub 2008 Mar 20. Brain Res. 2008. PMID: 18420182 Free PMC article.
References
-
- Baldassare, J.J., P.A. Henderson, D. Burns, C. Loomis, and G.J. Fisher. 1992. Translocation of protein kinase C isozymes in thrombin-stimulated human platelets. J. Biol. Chem. 267:15585–15590. - PubMed
-
- Bokkala, S., and S.K. Joseph. 1997. Angiotensin II-induced down-regulation of inositol trisphosphate receptors in WB rat liver epithelial cells. J. Biol. Chem. 272:12454–12461. - PubMed
-
- Codazzi, F., M.N. Teruel, and T. Meyer. 2001. Control of astrocyte Ca2+ oscillations and waves by oscillating translocation and activation of protein kinase C. Curr. Biol. 11:1089–1097. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

