Molecular analysis of maltotriose transport and utilization by Saccharomyces cerevisiae
- PMID: 12406721
- PMCID: PMC129873
- DOI: 10.1128/AEM.68.11.5326-5335.2002
Molecular analysis of maltotriose transport and utilization by Saccharomyces cerevisiae
Abstract
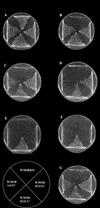
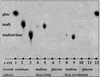
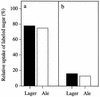
Efficient fermentation of maltotriose is a desired property of Saccharomyces cerevisiae for brewing. In a standard wort, maltotriose is the second most abundant sugar, and slower uptake leads to residual maltotriose in the finished product. The limiting factor of sugar metabolism is its transport, and there are conflicting reports on whether a specific maltotriose permease exists or whether the mechanisms responsible for maltose uptake also carry out maltotriose transport. In this study, radiolabeled maltotriose was used to show that overexpression of the maltose permease gene, MAL61, in an industrial yeast strain resulted in an increase in the rate of transport of maltotriose as well as maltose. A strain derived from W303-1A and lacking any maltose or maltotriose transporter but carrying a functional maltose transport activator (MAL63) was developed. By complementing this strain with permeases encoded by MAL31, MAL61, and AGT1, it was possible to measure their specific transport kinetics by using maltotriose and maltose. All three permeases were capable of high-affinity transport of maltotriose and of allowing growth of the strain on the sugar. Maltotriose utilization from the permease encoded by AGT1 was regulated by the same genetic mechanisms as those involving the maltose transcriptional activator. Competition studies carried out with two industrial strains, one not containing any homologue of AGT1, showed that maltose uptake and maltotriose uptake were competitive and that maltose was the preferred substrate. These results indicate that the presence of residual maltotriose in beer is not due to a genetic or physiological inability of yeast cells to utilize the sugar but rather to the lower affinity for maltotriose uptake in conjunction with deteriorating conditions present at the later stages of fermentation. Here we identify molecular mechanisms regulating the uptake of maltotriose and determine the role of each of the transporter genes in the cells.
Figures




References
-
- Attfield, P. V. 1997. Stress tolerance: the key to effective strains of industrial baker's yeast. Nat. Biotechnol. 15:1351-1357. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

