The A14-A43 heterodimer subunit in yeast RNA pol I and their relationship to Rpb4-Rpb7 pol II subunits
- PMID: 12407181
- PMCID: PMC137477
- DOI: 10.1073/pnas.232580799
The A14-A43 heterodimer subunit in yeast RNA pol I and their relationship to Rpb4-Rpb7 pol II subunits
Abstract
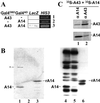
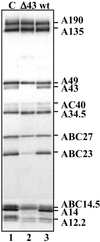
A43, an essential subunit of yeast RNA polymerase I (pol I), interacts with Rrn3, a class I general transcription factor required for rDNA transcription. The pol I-Rrn3 complex is the only form of enzyme competent for promoter-dependent transcription initiation. In this paper, using biochemical and genetic approaches, we demonstrate that the A43 polypeptide forms a stable heterodimer with the A14 pol I subunit and interacts with the common ABC23 subunit, the yeast counterpart of the omega subunit of bacterial RNA polymerase. We show by immunoelectronic microscopy that A43, ABC23, and A14 colocalize in the three-dimensional structure of the pol I, and we demonstrate that the presence of A43 is required for the stabilization of both A14 and ABC23 within the pol I. Because the N-terminal half of A43 is clearly related to the pol II Rpb7 subunit, we propose that the A43-A14 pair is likely the pol I counterpart of the Rpb7-Rpb4 heterodimer, although A14 distinguishes from Rpb4 by specific sequence and structure features. This hypothesis, combined with our structural data, suggests a new localization of Rpb7-Rpb4 subunits in the three-dimensional structure of yeast pol II.
Figures



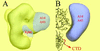

Similar articles
-
Structural and functional homology between the RNAP(I) subunits A14/A43 and the archaeal RNAP subunits E/F.Nucleic Acids Res. 2003 Aug 1;31(15):4391-400. doi: 10.1093/nar/gkg652. Nucleic Acids Res. 2003. PMID: 12888498 Free PMC article.
-
The fission yeast protein Ker1p is an ortholog of RNA polymerase I subunit A14 in Saccharomyces cerevisiae and is required for stable association of Rrn3p and RPA21 in RNA polymerase I.J Biol Chem. 2005 Mar 25;280(12):11467-74. doi: 10.1074/jbc.M411150200. Epub 2005 Jan 12. J Biol Chem. 2005. PMID: 15647272
-
The recruitment of RNA polymerase I on rDNA is mediated by the interaction of the A43 subunit with Rrn3.EMBO J. 2000 Oct 16;19(20):5473-82. doi: 10.1093/emboj/19.20.5473. EMBO J. 2000. PMID: 11032814 Free PMC article.
-
Rpb4 and Rpb7: subunits of RNA polymerase II and beyond.Trends Biochem Sci. 2004 Dec;29(12):674-81. doi: 10.1016/j.tibs.2004.10.007. Trends Biochem Sci. 2004. PMID: 15544954 Review.
-
Synthesis of the ribosomal RNA precursor in human cells: mechanisms, factors and regulation.Biol Chem. 2023 Jul 17;404(11-12):1003-1023. doi: 10.1515/hsz-2023-0214. Print 2023 Oct 26. Biol Chem. 2023. PMID: 37454246 Review.
Cited by
-
Genomewide recruitment analysis of Rpb4, a subunit of polymerase II in Saccharomyces cerevisiae, reveals its involvement in transcription elongation.Eukaryot Cell. 2008 Jun;7(6):1009-18. doi: 10.1128/EC.00057-08. Epub 2008 Apr 25. Eukaryot Cell. 2008. PMID: 18441121 Free PMC article.
-
Ribosomal RNA Transcription Machineries in Intestinal Protozoan Parasites: A Bioinformatic Analysis.Acta Parasitol. 2022 Dec;67(4):1788-1799. doi: 10.1007/s11686-022-00612-7. Epub 2022 Aug 27. Acta Parasitol. 2022. PMID: 36028726 Free PMC article.
-
Functional organization of the Rpb5 subunit shared by the three yeast RNA polymerases.Nucleic Acids Res. 2007;35(2):634-47. doi: 10.1093/nar/gkl686. Epub 2006 Dec 19. Nucleic Acids Res. 2007. PMID: 17179178 Free PMC article.
-
RNA pol II subunit RPB7 is required for RNA pol I-mediated transcription in Trypanosoma brucei.EMBO Rep. 2009 Mar;10(3):252-7. doi: 10.1038/embor.2008.244. Epub 2009 Jan 23. EMBO Rep. 2009. PMID: 19165144 Free PMC article.
-
Crystallization of RNA polymerase I subcomplex A14/A43 by iterative prediction, probing and removal of flexible regions.Acta Crystallogr Sect F Struct Biol Cryst Commun. 2008 May 1;64(Pt 5):413-8. doi: 10.1107/S174430910800972X. Epub 2008 Apr 24. Acta Crystallogr Sect F Struct Biol Cryst Commun. 2008. PMID: 18453714 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

