Structural basis for the oxidation of thiosulfate by a sulfur cycle enzyme
- PMID: 12411478
- PMCID: PMC131063
- DOI: 10.1093/emboj/cdf566
Structural basis for the oxidation of thiosulfate by a sulfur cycle enzyme
Abstract
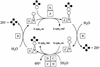
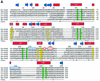
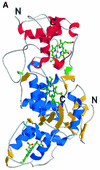
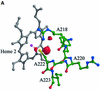
Reduced inorganic sulfur compounds are utilized by many bacteria as electron donors to photosynthetic or respiratory electron transport chains. This metabolism is a key component of the biogeochemical sulfur cycle. The SoxAX protein is a heterodimeric c-type cytochrome involved in thiosulfate oxidation. The crystal structures of SoxAX from the photosynthetic bacterium Rhodovulum sulfidophilum have been solved at 1.75 A resolution in the oxidized state and at 1.5 A resolution in the dithionite-reduced state, providing the first structural insights into the enzymatic oxidation of thiosulfate. The SoxAX active site contains a haem with unprecedented cysteine persulfide (cysteine sulfane) coordination. This unusual post-translational modification is also seen in sulfurtransferases such as rhodanese. Intriguingly, this enzyme shares further active site characteristics with SoxAX such as an adjacent conserved arginine residue and a strongly positive electrostatic potential. These similarities have allowed us to suggest a catalytic mechanism for enzymatic thiosulfate oxidation. The atomic coordinates and experimental structure factors have been deposited in the PDB with the accession codes 1H31, 1H32 and 1H33.
Figures






References
-
- Bartsch R.G. (1971) Cytochromes: bacterial. Methods Enzymol., 23, 344–363.
-
- Beinert H. (1983) Semi-micro methods for analysis of labile sulfide and of labile sulfide plus sulfane sulfur in unusually stable iron–sulfur proteins. Anal. Biochem., 131, 373–378. - PubMed
-
- Bordo D., Deriu,D., Colnaghi,R., Carpen,A., Pagani,S. and Bolognesi,M. (2000) The crystal structure of a sulfurtransferase from Azotobacter vinelandii highlights the evolutionary relationship between the rhodanese and phosphatase enzyme families. J. Mol. Biol., 298, 691–704. - PubMed
-
- Brune D.C. (1989) Sulfur oxidation by phototrophic bacteria. Biochim. Biophys. Acta, 975, 189–221. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

