Mapping of functional regions in the amino-terminal portion of the herpes simplex virus ICP27 regulatory protein: importance of the leucine-rich nuclear export signal and RGG Box RNA-binding domain
- PMID: 12414929
- PMCID: PMC136872
- DOI: 10.1128/jvi.76.23.11866-11879.2002
Mapping of functional regions in the amino-terminal portion of the herpes simplex virus ICP27 regulatory protein: importance of the leucine-rich nuclear export signal and RGG Box RNA-binding domain
Abstract
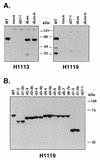
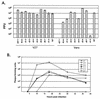
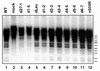
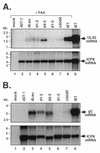
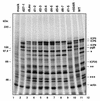
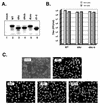
Infected-cell protein 27 (ICP27) is an essential herpes simplex virus type 1 (HSV-1) regulatory protein that activates a subset of viral delayed-early and late genes, at least in part through posttranscriptional mechanisms. Previous studies have shown that the amino (N)-terminal half of the protein contains important functional regions, including a leucine-rich nuclear export signal (NES). However, to date, the phenotype of an HSV-1 ICP27 NES mutant has not been reported. In this study, we engineered and characterized dLeu, an HSV-1 deletion mutant that specifically lacks ICP27's NES (amino acids 6 to 19). The phenotype of dLeu was analyzed alongside those of eight other ICP27 N-terminal deletion mutants. We found that in Vero cells, dLeu displays modest defects in viral gene expression and an approximately 100-fold reduction in the production of viral progeny. Unlike wild-type (WT) ICP27, which exhibits a cytoplasmic distribution in addition to its predominant nuclear localization, dLeu ICP27 is highly restricted to the cell nucleus. This strongly suggests that the N-terminal leucine-rich sequence functions as an NES during viral infection. Our analysis of dLeu and the other mutants has enabled us to genetically define the regions in the N-terminal 200 residues of ICP27 which are required for efficient viral growth in Vero cells. Only two regions appear to be important: (i) the leucine-rich NES and (ii) the RGG box RNA-binding domain, encoded by residues 139 to 153. A virus lacking the RGG box-encoding sequence, d4-5, has a phenotype similar to that of dLeu in that it displays modest defects in viral gene expression and grows poorly. Interestingly, deletion of both the NES and RGG box, as well as the sequences in between, is lethal. The resulting virus, d1-5, displays severe defects in viral gene expression and DNA synthesis and is unable to produce significant amounts of infectious progeny. Therefore, the N-terminal portion of ICP27 contains at least two functional domains which collectively are absolutely essential for viral infection.
Figures


References
-
- Blaho, J. A., C. Mitchell, and B. Roizman. 1994. An amino acid sequence shared by the herpes simplex virus 1 alpha regulatory proteins 0, 4, 22, and 27 predicts the nucleotidylylation of the UL21, UL31, UL47, and UL49 gene products. J. Biol. Chem. 269:17401-17410. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

