Effects of furosemide applied chronically to the round window: a model of metabolic presbyacusis
- PMID: 12417690
- PMCID: PMC6758027
- DOI: 10.1523/JNEUROSCI.22-21-09643.2002
Effects of furosemide applied chronically to the round window: a model of metabolic presbyacusis
Abstract
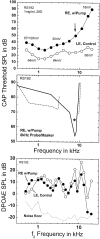
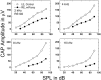
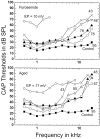
Hearing thresholds in elderly humans without a history of noise exposure commonly show a profile of a flat loss at low frequencies coupled with a loss that increases with frequency above approximately 2 kHz. This profile and the relatively robust distortion product otoacoustic emissions that are found in elderly subjects challenge the common belief that age-related hearing loss (presbyacusis) is based primarily on sensory-cell disorders. Here, we examine a model of presbyacusis wherein the endocochlear potential (EP) is reduced by means of furosemide applied chronically to one cochlea of a young gerbil. The model results in an EP that is reduced from 90 to approximately 60 mV, a value often seen in quiet-aged gerbils, with no concomitant loss of hair cells. Resulting measures of cochlear and neural function are quantitatively similar to those seen in aging gerbils and humans, e.g., a flat threshold loss at low frequencies with a high-frequency roll-off of approximately -8.4 dB/octave. The effect of the EP on neural thresholds can be parsimoniously explained by the known gain characteristics of the cochlear amplifier as a function of cochlear location: in the apex, amplification is limited to approximately 20 dB, whereas in the base, the gain can be as high as 60 dB. At high frequencies, amplification is directly proportional to the EP on an approximately 1 dB/mV basis. This model suggests that the primary factor in true age-related hearing loss is an energy-starved cochlear amplifier that results in a specific audiogram profile.
Figures




References
-
- Bhattacharyya TK, Dayal VS. Age-related cochlear hair cell loss in the chinchilla. Ann Otol Rhinol Laryngol. 1985;94:75–80. - PubMed
-
- Boettcher FA, Schmiedt RA. Distortion-product otoacoustic emissions in Mongolian gerbils with resistance to noise-induced hearing loss. J Acoust Soc Am. 1995;98:3215–3222. - PubMed
-
- Castor X, Veuillet E, Morgon A, Collet L. Influence of aging on cochlear micromechanical properties and on the medial olivocochlear system in humans. Hear Res. 1994;77:1–8. - PubMed
-
- Chamberlain SC. Neuroanatomical aspects of the gerbil's inner ear: light microscopic observations. J Comp Neurol. 1977;171:193–204. - PubMed
-
- Cooper NP, Rhode WS. Mechanical responses to two-tone distortion products in the apical and basal turns of the mammalian cochlea. J Neurophysiol. 1997;78:261–270. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
