Salt stress inhibits the repair of photodamaged photosystem II by suppressing the transcription and translation of psbA genes in synechocystis
- PMID: 12428009
- PMCID: PMC166663
- DOI: 10.1104/pp.011114
Salt stress inhibits the repair of photodamaged photosystem II by suppressing the transcription and translation of psbA genes in synechocystis
Abstract
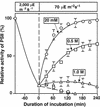
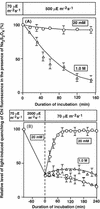
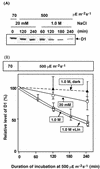
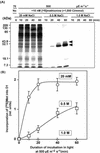
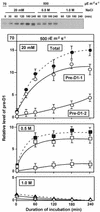
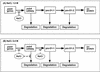
Light stress and salt stress are major environmental factors that limit the efficiency of photosynthesis. However, we have found that the effects of light and salt stress on photosystem II (PSII) in the cyanobacterium Synechocystis sp. PCC 6803 are completely different. Strong light induced photodamage to PSII, whereas salt stress inhibited the repair of the photodamaged PSII and did not accelerate damage to PSII directly. The combination of light and salt stress appeared to inactivate PSII very rapidly as a consequence of their synergistic effects. Radioactive labeling of cells revealed that salt stress inhibited the synthesis of proteins de novo and, in particular, the synthesis of the D1 protein. Northern- and western-blotting analyses demonstrated that salt stress inhibited the transcription and the translation of psbA genes, which encode D1 protein. DNA microarray analysis indicated that the light-induced expression of various genes was suppressed by salt stress. Thus, our results suggest that salt stress inhibits the repair of PSII via suppression of the activities of the transcriptional and translational machinery.
Figures




References
-
- Allakhverdiev SI, Klimov VV, Ladygin VG. Photoreduction of pheophytin in photosystem II reaction centers of intact cells of green algae and cyanobacteria under anaerobic conditions. Biofizika (Moscow) 1988;33:442–447. - PubMed
-
- Arnon DI, McSwain BD, Tsujimoto HY, Wada K. Photochemical activity and components of membrane preparations from blue-green algae: coexistence of two photosystems in relation to chlorophyll a and removal of phycocyanin. Biochim Biophys Acta. 1974;357:231–245. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

