Colony shape as a genetic trait in the pattern-forming Bacillus mycoides
- PMID: 12429070
- PMCID: PMC138795
- DOI: 10.1186/1471-2180-2-33
Colony shape as a genetic trait in the pattern-forming Bacillus mycoides
Abstract
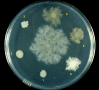
Background: Bacillus mycoides Flügge, a Gram-positive, non-motile soil bacterium assigned to Bacillus cereus group, grows on agar as chains of cells linked end to end, forming radial filaments curving clock- or counter-clockwise (SIN or DX morphotypes). The molecular mechanism causing asymmetric curving is not known: our working hypothesis considers regulation of filamentous growth as the prerequisite for these morphotypes.
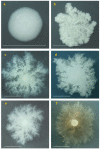
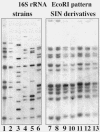
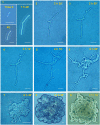
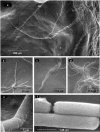
Results: SIN and DX strains isolated from the environment were classified as B. mycoides by biochemical and molecular biology tests. Growth on agar of different hardness and nutrient concentration did not abolish colony patterns, nor was conversion between SIN and DX morphotypes ever noticed. A number of morphotype mutants, all originating from one SIN strain, were obtained. Some lost turn direction becoming fluffy, others became round and compact. All mutants lost wild type tight aggregation in liquid culture. Growth on agar was followed by microscopy, exploring the process of colony formation and details of cell divisions. A region of the dcw (division cell wall) cluster, including ftsQ, ftsA, ftsZ and murC, was sequenced in DX and SIN strains as a basis for studying cell division. This confirmed the relatedness of DX and SIN strains to the B. cereus group.
Conclusions: DX and SIN asymmetric morphotypes stem from a close but not identical genomic context. Asymmetry is established early during growth on agar. Wild type bacilli construct mostly uninterrupted filaments with cells dividing at the free ends: they "walk" longer distances compared to mutants, where enhanced frequency of cell separation produces new growing edges resulting in round compact colonies.
Figures







References
-
- Shapiro JA. The significance of bacterial colony patterns. BioEssays. 1995;17:597–607. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

