The colon-selective spasmolytic otilonium bromide inhibits muscarinic M(3) receptor-coupled calcium signals in isolated human colonic crypts
- PMID: 12429587
- PMCID: PMC1573573
- DOI: 10.1038/sj.bjp.0704942
The colon-selective spasmolytic otilonium bromide inhibits muscarinic M(3) receptor-coupled calcium signals in isolated human colonic crypts
Abstract
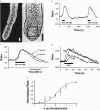
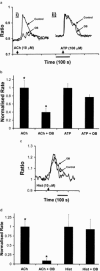
1. Otilonium bromide (OB) is a smooth muscle relaxant used in the treatment of irritable bowel syndrome. Otilonium bromide has been shown to interfere with the mobilization of calcium in intestinal smooth muscle, but the effects on other intestinal tissues have not been investigated. We identified the muscarinic receptor subtype coupled to calcium signals in colonic crypt derived from the human colonic epithelium and evaluated the inhibitory effects of OB. 2. Calcium signals were monitored by fluorescence imaging of isolated human colonic crypts and Chinese hamster ovary cells stably expressing the cloned human muscarinic M(3) receptor subtype (CHO-M(3)). Colonic crypt receptor expression was investigated by pharmacological and immunohistochemical techniques. 3. The secretagogue acetylcholine (ACh) stimulated calcium mobilization from intracellular calcium stores at the base of human colonic crypts with an EC(50) of 14 micro M. The muscarinic receptor antagonists 4-DAMP, AF-DX 384, pirenzepine and methroctamine inhibited the ACh-induced calcium signal with the following respective IC(50) (pK(b)) values: 0.78 nM (9.1), 69 nM (7.2), 128 nM (7.1), and 2510 nM (5.8). 4. Immunohistochemical analyses of muscarinic receptor expression demonstrated the presence of M(3) receptor subtype expression at the crypt-base. 5. Otilonium bromide inhibited the generation of ACh-induced calcium signals in a dose dependent manner (IC(50)=880 nM). 6. In CHO-M(3) cells, OB inhibited calcium signals induced by ACh, but not ATP. In addition, OB did not inhibit histamine-induced colonic crypt calcium signals. 7. The present studies have demonstrated that OB inhibited M(3) receptor-coupled calcium signals in human colonic crypts and CHO-M(3) cells, but not those induced by stimulation of other endogenous receptor types. We propose that the M(3) receptor-coupled calcium signalling pathway is directly targeted by OB at the level of the colonic epithelium, suggestive of an anti-secretory action in IBS patients suffering with diarrhoea.
Figures



Similar articles
-
Effect of otilonium bromide on contractile patterns in the human sigmoid colon.Neurogastroenterol Motil. 2010 Jun;22(6):e180-91. doi: 10.1111/j.1365-2982.2010.01495.x. Epub 2010 Mar 31. Neurogastroenterol Motil. 2010. PMID: 20367799
-
Mechanisms of action of otilonium bromide (OB) in human cultured smooth muscle cells and rat colonic strips.Neurogastroenterol Motil. 2013 Dec;25(12):e803-12. doi: 10.1111/nmo.12206. Epub 2013 Aug 14. Neurogastroenterol Motil. 2013. PMID: 23941257
-
Muscarinic--but not nicotinic--acetylcholine receptors mediate a nitric oxide-dependent dilation in brain cortical arterioles: a possible role for the M5 receptor subtype.J Cereb Blood Flow Metab. 2000 Feb;20(2):298-305. doi: 10.1097/00004647-200002000-00011. J Cereb Blood Flow Metab. 2000. PMID: 10698067
-
Pharmacological studies on otilonium bromide.Ital J Gastroenterol. 1991 Nov;23(8 Suppl 1):56-9. Ital J Gastroenterol. 1991. PMID: 1661629 Review.
-
Otilonium bromide: a selective spasmolytic for the gastrointestinal tract.J Int Med Res. 1999 Sep-Oct;27(5):207-22. doi: 10.1177/030006059902700501. J Int Med Res. 1999. PMID: 10689627 Review.
Cited by
-
Colonic smooth muscle cells and colonic motility patterns as a target for irritable bowel syndrome therapy: mechanisms of action of otilonium bromide.Therap Adv Gastroenterol. 2014 Jul;7(4):156-66. doi: 10.1177/1756283X14525250. Therap Adv Gastroenterol. 2014. PMID: 25057296 Free PMC article. Review.
-
Recent advances in pharmacological treatment of irritable bowel syndrome.World J Gastroenterol. 2014 Jul 21;20(27):8867-85. doi: 10.3748/wjg.v20.i27.8867. World J Gastroenterol. 2014. PMID: 25083060 Free PMC article. Review.
-
Efficacy of Solifenacin on Irritable Bowel Syndrome With Diarrhea: Open-label Prospective Pilot Trial.J Neurogastroenterol Motil. 2012 Jul;18(3):317-23. doi: 10.5056/jnm.2012.18.3.317. Epub 2012 Jul 10. J Neurogastroenterol Motil. 2012. PMID: 22837880 Free PMC article.
-
The evaluation of otilonium bromide treatment in asian patients with irritable bowel syndrome.J Neurogastroenterol Motil. 2011 Oct;17(4):402-10. doi: 10.5056/jnm.2011.17.4.402. Epub 2011 Oct 31. J Neurogastroenterol Motil. 2011. PMID: 22148110 Free PMC article.
-
Abolition of Ca2+-mediated intestinal anion secretion and increased stool dehydration in mice lacking the intermediate conductance Ca2+-dependent K+ channel Kcnn4.J Physiol. 2007 Sep 1;583(Pt 2):705-17. doi: 10.1113/jphysiol.2007.134387. Epub 2007 Jun 21. J Physiol. 2007. PMID: 17584847 Free PMC article.
References
-
- BATTAGLIA G., MORSELLI-LABATE A.M., CAMARRI E., FRANCAVILLA A., DE MARCO F., MASTROPAOLO G., NACCARATO R. Otilonium bromide in irritable bowel syndrome: a double-blind, placebo-controlled, 15-week study. Aliment. Pharmacol. Ther. 1998;12:1003–1010. - PubMed
-
- BARRETT K.E., KEELY S.J. Chloride secretion by the intestinal epithelium: molecular basis and regulatory aspects. Annu. Rev. Physiol. 2000;62:535–572. - PubMed
-
- BERRIDGE MJ. Inositol trisphosphate and calcium signalling. Nature. 1993;361:315–325. - PubMed
-
- BUCKLEY N.J., BONNER T.I., BUCKLEY C.M., BRANN M.R. Antagonist binding properties of five cloned muscarinic receptors expressed in CHO-K1 cells. Mol. Pharmacol. 1989;35:469–476. - PubMed
-
- CHANG W.W., LEBLOND C.P. Renewal of the epithelium in the descending colon of the mouse. I. Presence of three cell populations:vacuolated-columnar, mucous and argentaffin. Am. J. Anat. 1971;31:73–99. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

