Excitation- and beta(2)-agonist-induced activation of the Na(+)-K(+) pump in rat soleus muscle
- PMID: 12433963
- PMCID: PMC2290663
- DOI: 10.1113/jphysiol.2002.023325
Excitation- and beta(2)-agonist-induced activation of the Na(+)-K(+) pump in rat soleus muscle
Abstract
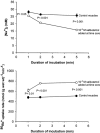
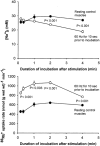
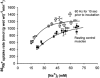
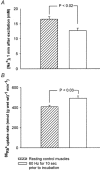
In rat skeletal muscle, Na(+)-K(+) pump activity increases dramatically in response to excitation (up to 20-fold) or beta(2)-agonists (2-fold), leading to a reduction in intracellular Na(+). This study examines the time course of these effects and whether they are due to an increased affinity of the Na(+)-K(+) pump for intracellular Na(+). Isolated rat soleus muscles were incubated at 30 (o)C in Krebs-Ringer bicarbonate buffer. The effects of direct electrical stimulation on (86)Rb(+) uptake rate and intracellular Na(+) concentration ([Na(+)](i)) were characterized in the subsequent recovery phase. [Na(+)](i) was varied using monensin or buffers with low Na(+). In the [Na(+)](i) range 21-69 mM, both the beta(2)-agonist salbutamol and electrical stimulation produced a left shift of the curves relating (86)Rb(+) uptake rate to [Na(+)](i). In the first 10 s after 1 or 10 s pulse trains of 60 Hz, [Na(+)](i) showed no increase, but (86)Rb(+) uptake rate increased by 22 and 86 %, respectively. Muscles excited in Na(+)-free Li(+)-substituted buffer and subsequently allowed to rest in standard buffer also showed a significant increase in (86)Rb(+) uptake rate and decrease in [Na(+)](i). Na(+) loading induced by monensin or electroporation also stimulated (86)Rb(+) uptake rate but, contrary to excitation, increased [Na(+)](i). The increase in the rate of (86)Rb(+) uptake elicited by electrical stimulation was abolished by ouabain, but not by bumetanide. The results indicate that excitation (like salbutamol) induces a rapid increase in the affinity of the Na(+)-K(+) pump for intracellular Na(+). This leads to a Na(+)-K(+) pump activation that does not require Na(+) influx, but possibly the generation of action potentials. This improves restoration of the Na(+)-K(+) homeostasis during work and optimizes excitability and contractile performance of the working muscle.
Figures



References
-
- Akera T, Bennet RT, Olgaard MK, Brody TM. Cardiac Na+, K+-adenosine triphosphatase inhibition by ouabain and myocardial sodium: A computer simulation. Journal of Pharmacology and Experimental Therapeutics. 1976;199:287–297. - PubMed
-
- Bigland-Ritchie B, Jones DA, Woods JJ. Excitation frequency and muscle fatigue: electrical responses during human voluntary and stimulated contractions. Experimental Neurology. 1979;64:414–427. - PubMed
-
- Buchanan R, Nielsen OB, Clausen T. Excitation- and β2-agonist-induced activation of the Na+, K+-pump in skeletal muscle. Acta Physiologica Scandinavica. 2002;174:A182.
-
- Clausen T, Everts ME. K+ induced inhibition of contractile force in rat skeletal muscle: role of active Na+–K+ transport. American Journal of Physiology. 1991;261:799–807. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

