T-cell responses to the Mycobacterium tuberculosis-specific antigen ESAT-6 in Brazilian tuberculosis patients
- PMID: 12438345
- PMCID: PMC132944
- DOI: 10.1128/IAI.70.12.6707-6714.2002
T-cell responses to the Mycobacterium tuberculosis-specific antigen ESAT-6 in Brazilian tuberculosis patients
Abstract
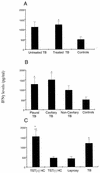
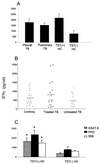
The Mycobacterium tuberculosis-specific ESAT-6 antigen induces highly potent T-cell responses and production of gamma interferon (IFN-gamma), which play a critical role in protective cell-mediated immunity against tuberculosis (TB). In the present study, IFN-gamma secretion by peripheral blood mononuclear cells (PBMCs) in response to M. tuberculosis ESAT-6 in Brazilian TB patients was investigated in relation to clinical disease types, such as pleurisy and cavitary pulmonary TB. Leprosy patients, patients with pulmonary diseases other than TB, and healthy donors were assayed as control groups. Sixty percent of the TB patients indeed recognized M. tuberculosis ESAT-6, as did 50% of the leprosy patients and 60% of the non-TB controls. Nevertheless, the levels of IFN-gamma in response to the antigen ESAT, but not to antigen 85B (Ag85B) and purified protein derivative (PPD), were significantly lower in controls than in patients with treated TB or pleural or cavitary TB. Moreover, according to Mycobacterium bovis BCG vaccination status, only 59% of the vaccinated TB patients responded to ESAT in vitro, whereas 100% of them responded to PPD. Both CD4 and CD8 T cells were able to release IFN-gamma in response to ESAT. The present data demonstrate the specificity of ESAT-6 of M. tuberculosis and its ability to discriminate TB patients from controls, including leprosy patients. However, to obtain specificity, it is necessary to include quantitative IFN-gamma production in response to the antigen as well, and this might limit the use of ESAT-6-based immunodiagnosis of M. tuberculosis infection in an area of TB endemicity.
Figures

Comment in
-
Are cultures needed to enroll tuberculosis patients?Infect Immun. 2003 Dec;71(12):7236; author reply 7236-7. doi: 10.1128/IAI.71.12.7236-7237.2003. Infect Immun. 2003. PMID: 14638825 Free PMC article. No abstract available.
Similar articles
-
ESAT-6-dependent cytosolic pattern recognition drives noncognate tuberculosis control in vivo.J Clin Invest. 2016 Jun 1;126(6):2109-22. doi: 10.1172/JCI84978. Epub 2016 Apr 25. J Clin Invest. 2016. PMID: 27111234 Free PMC article.
-
Cellular responses to MPT-51, GlcB and ESAT-6 among MDR-TB and active tuberculosis patients in Brazil.Tuberculosis (Edinb). 2008 Sep;88(5):474-81. doi: 10.1016/j.tube.2008.06.002. Epub 2008 Jul 26. Tuberculosis (Edinb). 2008. PMID: 18676203
-
Tuberculin-purified protein derivative-, MPT-64-, and ESAT-6-stimulated gamma interferon responses in medical students before and after Mycobacterium bovis BCG vaccination and in patients with tuberculosis.Clin Diagn Lab Immunol. 1999 Nov;6(6):934-7. doi: 10.1128/CDLI.6.6.934-937.1999. Clin Diagn Lab Immunol. 1999. PMID: 10548589 Free PMC article. Clinical Trial.
-
[Evolution of IGRA researches].Kekkaku. 2008 Sep;83(9):641-52. Kekkaku. 2008. PMID: 18979999 Review. Japanese.
-
Th22 response induced by Mycobacterium tuberculosis strains is closely related to severity of pulmonary lesions and bacillary load in patients with multi-drug-resistant tuberculosis.Clin Exp Immunol. 2021 Feb;203(2):267-280. doi: 10.1111/cei.13544. Epub 2020 Nov 18. Clin Exp Immunol. 2021. PMID: 33128773 Free PMC article. Review.
Cited by
-
T-cell and serological responses to Erp, an exported Mycobacterium tuberculosis protein, in tuberculosis patients and healthy individuals.BMC Infect Dis. 2007 Jul 26;7:83. doi: 10.1186/1471-2334-7-83. BMC Infect Dis. 2007. PMID: 17655752 Free PMC article.
-
Identification of early secretory antigen target-6 epitopes for the immunodiagnosis of active tuberculosis.Mol Med. 2003 Mar-Apr;9(3-4):105-11. Mol Med. 2003. PMID: 12865946 Free PMC article.
-
Are cultures needed to enroll tuberculosis patients?Infect Immun. 2003 Dec;71(12):7236; author reply 7236-7. doi: 10.1128/IAI.71.12.7236-7237.2003. Infect Immun. 2003. PMID: 14638825 Free PMC article. No abstract available.
-
Unique model of dormant infection for tuberculosis vaccine development.Clin Vaccine Immunol. 2006 Sep;13(9):1014-21. doi: 10.1128/CVI.00120-06. Clin Vaccine Immunol. 2006. PMID: 16960113 Free PMC article.
-
Rv0315, a novel immunostimulatory antigen of Mycobacterium tuberculosis, activates dendritic cells and drives Th1 immune responses.J Mol Med (Berl). 2012 Mar;90(3):285-98. doi: 10.1007/s00109-011-0819-2. Epub 2011 Oct 13. J Mol Med (Berl). 2012. PMID: 21993523
References
-
- Andersen, P. 1994. The T cell response to secreted antigens of M. tuberculosis. Immunobiol. 191:537-547. - PubMed
-
- Arend, S. M., P. Andersen, K. E. van Meijaarden, R. L. Skjot, Y. W. Subronto, J. T. van Dissel, and T. H. Ottenhoff. 2000. Detection of active tuberculosis infection by T cell responses to early-secreted antigenic target 6kDa protein and culture filtrate protein 10. J. Infect. Dis. 181:1850-1854. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

