The unfolded protein response modulates disease severity in Pelizaeus-Merzbacher disease
- PMID: 12441049
- PMCID: PMC4603660
- DOI: 10.1016/s0896-6273(02)01045-0
The unfolded protein response modulates disease severity in Pelizaeus-Merzbacher disease
Abstract
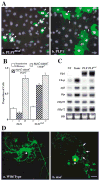
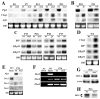
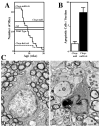
The unfolded protein response (UPR) is a eukaryotic signaling pathway linking protein flux through the endoplasmic reticulum to transcription and translational repression. Herein, we demonstrate UPR activation in the leukodystrophy Pelizaeus-Merzbacher disease (PMD) as well as in three mouse models of this disease and transfected fibroblasts expressing mutant protein. The CHOP protein, widely known as a proapoptotic transcription factor, modulates pathogenesis in the mouse models of PMD; however, this protein exhibits antiapoptotic activity. Together, these data show that the UPR has the potential to modulate disease severity in many cells expressing mutant secretory pathway proteins. Thus, PMD represents the first member of a novel class of disparate degenerative diseases for which UPR activation and signaling is the common pathogenic mechanism.
Figures





References
-
- Allison J, Campbell IL, Morahan G, Mandel TE, Harrison LC, Miller JFAP. Diabetes in transgenic mice resulting from over-expression of class I histocompatibility molecules in pancreatic β cells. Nature. 1988;333:529–533. - PubMed
-
- Aridor M, Balch WE. Integration of endoplasmic reticulum signaling in health and disease. Nat Med. 1999;5:745–751. - PubMed
-
- Baerwald KD, Corbin JG, Popko B. Major histocompatibility complex heavy chain accumulation in the endoplasmic reticulum of oligodendrocytes results in myelin abnormalities. J Neurosci Res. 2000;59:160–169. - PubMed
-
- Chirgwin J, Przybyla A, MacDonald R, Rutter W. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry. 1979;18:5294–5299. - PubMed
-
- Duncan ID. Dissection of the phenotype and genotype of the X-linked myelin mutants. In: Duncan ID, Skoff RP, Colman DR, editors. Myelination and Dysmyelination. New York: New York Academy of Sciences; 1990. pp. 110–121. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

