Delayed rectifier K+ currents, IK, are encoded by Kv2 alpha-subunits and regulate tonic firing in mammalian sympathetic neurons
- PMID: 12451110
- PMCID: PMC6758768
- DOI: 10.1523/JNEUROSCI.22-23-10094.2002
Delayed rectifier K+ currents, IK, are encoded by Kv2 alpha-subunits and regulate tonic firing in mammalian sympathetic neurons
Abstract
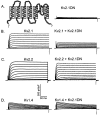
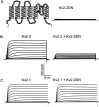
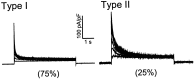
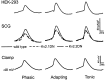
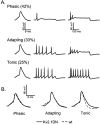
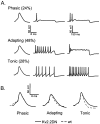
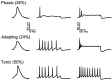
Previous studies have revealed the presence of four kinetically distinct voltage-gated K+ currents, I(Af), I(As), I(K), and I(SS), in rat superior cervical ganglion (SCG) neurons and demonstrated that I(K) and I(SS) are expressed in all cells, whereas I(Af) and I(As) are differentially distributed. Previous studies have also revealed the presence of distinct components of I(Af) encoded by alpha-subunits of the Kv1 and Kv4 subfamilies. In the experiments described here, pore mutants of Kv2.1 (Kv2.1W365C/Y380T) and Kv2.2 (Kv2.2W373C/Y388T) that function as Kv2 subfamily-specific dominant negatives (Kv2.1DN and Kv2.2DN) were generated to probe the functional role(s) of Kv2 alpha-subunits. Expression of Kv2.1DN or Kv2.2DN in human embryonic kidney-293 cells selectively attenuates Kv2.1- or Kv2.2-encoded K+ currents, respectively. Using the Biolistics Gene Gun, cDNA constructs encoding either Kv2.1DN or Kv2.2DN [and enhanced green fluorescent protein (EGFP)] were introduced into SCG neurons. Whole-cell recordings from EGFP-positive Kv2.1DN or Kv2.2DN-expressing cells revealed selective decreases in I(K). Coexpression of Kv2.1DN and Kv2.2DN eliminates I(K) in most (75%) SCG cells and, in the remaining (25%) cells, I(K) density is reduced. Together with biochemical data revealing that Kv2.1 and Kv2.2 alpha-subunits do not associate in rat SCGs, these results suggest that Kv2.1 and Kv2.2 form distinct populations of I(K) channels, and that Kv2 alpha-subunits underlie (most of) I(K) in SCG neurons. Similar to wild-type cells, phasic, adapting, and tonic firing patterns are evident in SCG cells expressing Kv2.1DN or Kv2.2DN, although action potential durations in tonic cells are prolonged. Expression of Kv2.2DN also results in membrane depolarization, suggesting that Kv2.1- and Kv2.2-encoded I(K) channels play distinct roles in regulating the excitability of SCG neurons.
Figures



Similar articles
-
Molecular heterogeneity of the voltage-gated fast transient outward K+ current, I(Af), in mammalian neurons.J Neurosci. 2001 Oct 15;21(20):8004-14. doi: 10.1523/JNEUROSCI.21-20-08004.2001. J Neurosci. 2001. PMID: 11588173 Free PMC article.
-
Elimination of the fast transient in superior cervical ganglion neurons with expression of KV4.2W362F: molecular dissection of IA.J Neurosci. 2000 Jul 15;20(14):5191-9. doi: 10.1523/JNEUROSCI.20-14-05191.2000. J Neurosci. 2000. PMID: 10884302 Free PMC article.
-
Attenuation of the slow component of delayed rectification, action potential prolongation, and triggered activity in mice expressing a dominant-negative Kv2 alpha subunit.Circ Res. 1999 Oct 1;85(7):623-33. doi: 10.1161/01.res.85.7.623. Circ Res. 1999. PMID: 10506487
-
Genes responsible for native depolarization-activated K+ currents in neurons.Neurosci Res. 2002 Jan;42(1):7-14. doi: 10.1016/s0168-0102(01)00305-4. Neurosci Res. 2002. PMID: 11814604 Review.
-
Regulation of voltage-gated K+ channel expression in the developing mammalian myocardium.J Neurobiol. 1998 Oct;37(1):37-59. doi: 10.1002/(sici)1097-4695(199810)37:1<37::aid-neu4>3.0.co;2-9. J Neurobiol. 1998. PMID: 9777731 Review.
Cited by
-
The C-terminus of neuronal Kv2.1 channels is required for channel localization and targeting but not for NMDA-receptor-mediated regulation of channel function.Neuroscience. 2012 Aug 16;217:56-66. doi: 10.1016/j.neuroscience.2012.04.054. Epub 2012 Apr 30. Neuroscience. 2012. PMID: 22554782 Free PMC article.
-
Characterization of the Kv channels of mouse carotid body chemoreceptor cells and their role in oxygen sensing.J Physiol. 2004 Jun 1;557(Pt 2):457-71. doi: 10.1113/jphysiol.2004.062281. Epub 2004 Mar 19. J Physiol. 2004. PMID: 15034123 Free PMC article.
-
MinK-related peptide 2 modulates Kv2.1 and Kv3.1 potassium channels in mammalian brain.J Neurosci. 2003 Sep 3;23(22):8077-91. doi: 10.1523/JNEUROSCI.23-22-08077.2003. J Neurosci. 2003. PMID: 12954870 Free PMC article.
-
Electrical remodelling maintains firing properties in cortical pyramidal neurons lacking KCND2-encoded A-type K+ currents.J Physiol. 2008 Mar 15;586(6):1565-79. doi: 10.1113/jphysiol.2007.146597. Epub 2008 Jan 10. J Physiol. 2008. PMID: 18187474 Free PMC article.
-
Whole cell recording from an organotypic slice preparation of neocortex.J Vis Exp. 2011 Jun 3;(52):2600. doi: 10.3791/2600. J Vis Exp. 2011. PMID: 21673642 Free PMC article.
References
-
- An WF, Bowlby MR, Betty M, Cao J, Ling H-P, Mendoza G, Hinson JW, Mattson KI, Strassle BW, Trimmer JS, Rhodes KJ. Modulation of A-type potassium channels by a family of calcium sensors. Nature. 2000;403:553–556. - PubMed
-
- Barry DM, Trimmer JS, Merlie JP, Nerbonne JM. Differential expression of voltage-gated K+ channel subunits in adult rat heart. Relation to functional K+ channels. Circ Res. 1995;77:361–369. - PubMed
-
- Barry DM, Xu H, Schuessler RB, Nerbonne JM. Functional knock-out of the transient outward current, long-QT syndrome, and cardiac remodeling in mice expressing a dominant-negative Kv4 α-subunit. Circ Res. 1998;83:560–567. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
