A role for the redox site in the modulation of the NMDA receptor by light
- PMID: 12456823
- PMCID: PMC2290695
- DOI: 10.1113/jphysiol.2002.032755
A role for the redox site in the modulation of the NMDA receptor by light
Abstract
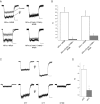
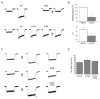
Light has been shown to modulate NMDA receptor function. In this study, we have performed experiments aimed at elucidating the putative site of action of light within the receptor structure. Whole-cell recordings were performed in Chinese hamster ovary cells expressing various combinations of NMDA receptor subunits. Although there was no apparent difference in the actions of light between wild-type NR1-NR2A and NR1-NR2B subunit configurations, the light enhancement of NMDA-induced currents was either completely abolished or substantially diminished in the redox site mutants NR1a (C744A, C798A)-NR2B and NR1a (C744A, C798A)-NR2A. Further studies demonstrated that chemical reduction of NR1a-NR2B NMDA receptors decreased its sensitivity to light. In addition, sodium (2-sulfonatoethyl) methanethiosulfonate (MTSES), used to irreversibly bind free sulfhydryl groups and inactivate the redox site, abolished the effects of light on wild-type receptors. In contrast, no free sulfhydryls were available for MTSES following light stimulation, suggesting that light itself could not reduce the redox modulatory site. Our results suggest that a functionally intact, oxidized redox site is necessary for light-induced potentiation. Hence, light and redox modulation of the NMDA receptor may share a common intramolecular pathway for altering the function of this ion channel.
Figures
Similar articles
-
A critical role of the N-methyl-D-aspartate (NMDA) receptor subunit (NR) 2A in the expression of redox sensitivity of NR1/NR2A recombinant NMDA receptors.J Pharmacol Exp Ther. 1999 Nov;291(2):785-92. J Pharmacol Exp Ther. 1999. PMID: 10525101
-
Functional consequences of NR2 subunit composition in single recombinant N-methyl-D-aspartate receptors.Proc Natl Acad Sci U S A. 1997 Sep 30;94(20):11019-24. doi: 10.1073/pnas.94.20.11019. Proc Natl Acad Sci U S A. 1997. PMID: 9380752 Free PMC article.
-
Lack of interaction between nitric oxide and the redox modulatory site of the NMDA receptor.Br J Pharmacol. 1999 Jan;126(1):296-300. doi: 10.1038/sj.bjp.0702295. Br J Pharmacol. 1999. PMID: 10051148 Free PMC article.
-
NMDA receptor redox sites: are they targets for selective neuronal protection?Trends Pharmacol Sci. 1995 Nov;16(11):368-74. doi: 10.1016/s0165-6147(00)89077-x. Trends Pharmacol Sci. 1995. PMID: 8578605 Review.
-
Redox modulation of the NMDA receptor.Cell Mol Life Sci. 2000 Oct;57(11):1535-41. doi: 10.1007/pl00000638. Cell Mol Life Sci. 2000. PMID: 11092448 Free PMC article. Review.
Cited by
-
The Redox Biology of Excitotoxic Processes: The NMDA Receptor, TOPA Quinone, and the Oxidative Liberation of Intracellular Zinc.Front Neurosci. 2020 Jul 24;14:778. doi: 10.3389/fnins.2020.00778. eCollection 2020. Front Neurosci. 2020. PMID: 32792905 Free PMC article. Review.
-
Reducing agents sensitize C-type nociceptors by relieving high-affinity zinc inhibition of T-type calcium channels.J Neurosci. 2007 Aug 1;27(31):8250-60. doi: 10.1523/JNEUROSCI.1800-07.2007. J Neurosci. 2007. PMID: 17670971 Free PMC article.
-
NMDA potentiation by visible light in the presence of a fluorescent neurosteroid analogue.J Physiol. 2009 Jun 15;587(Pt 12):2937-47. doi: 10.1113/jphysiol.2009.172700. Epub 2009 Apr 29. J Physiol. 2009. PMID: 19403611 Free PMC article.
-
Photodynamic effects of steroid-conjugated fluorophores on GABAA receptors.Mol Pharmacol. 2009 Oct;76(4):754-65. doi: 10.1124/mol.109.057687. Epub 2009 Jul 13. Mol Pharmacol. 2009. PMID: 19596835 Free PMC article.
References
-
- Aizenman E, Lipton SA, Loring RH. Selective modulation of NMDA responses by reduction and oxidation. Neuron. 1989;2:1257–1263. - PubMed
-
- Akabas MH, Stauffer DA, Xu M, Karlin A. Acetylcholine receptor channel structure probed in cysteine-substitution mutants. Science. 1992;258:307–310. - PubMed
-
- Boeckman FA, Aizenman E. Pharmacological properties of acquired excitotoxicity in Chinese hamster ovary cells transfected with N-methyl-d-aspartate receptor subunits. Journal of Pharmacology and Experimental Therapeutics. 1996;279:515–523. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

