Single-cell epithelial defects close rapidly by an actinomyosin purse string mechanism with functional tight junctions
- PMID: 12456828
- PMCID: PMC2290693
- DOI: 10.1113/jphysiol.2002.031161
Single-cell epithelial defects close rapidly by an actinomyosin purse string mechanism with functional tight junctions
Abstract
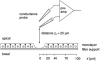
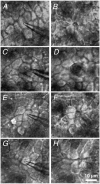
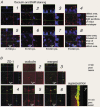
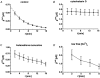
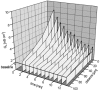
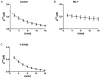
Restitution of single-cell defects, a frequent event in epithelia with high turnover, is poorly understood. Morphological and functional changes were recorded, using intravital time-lapse video microscopy, confocal fluorescence microscopy, and conductance scanning techniques. After artificial single-cell loss from an HT-29/B6 colonic cell monolayer, the basal ends of adjacent cells extended. Concurrently, the local conductive leak associated with the defect sealed with an exponential time course (from 0.48 +/- 0.05 microS 2 min post lesion to 0.17 +/- 0.02 microS 8 min post lesion, n = 17). Between 3 and 10 min post lesion, a band of actin arose around the gap, which colocalized with a ring of ZO-1 and occludin. Hence, tight junction proteins bound to the actin band facing the gap, and competent tight junctions assembled in the adjoining cell membranes. Closure and sealing were inhibited when actin polymerization was blocked by cytochalasin D, delayed following decrease of myosin-ATPase activity by butanedione monoxime, and blocked after myosin light chain kinase inhibition by ML-7. The Rho-associated protein kinase inhibitor Y-27632 did not affect restitution. After loosening of intercellular contacts in low Ca(2+) Ringer solution, the time course of restitution was not significantly altered. Albeit epithelial conductivity was 12-fold higher in low Ca(2+) Ringer solution than in controls, under both conditions the repaired epithelium assumed the same conductivity as distant intact epithelium. In conclusion, epithelial restitution of single-cell defects comprises rapid closure by an actinomyosin 'purse-string' mechanism and simultaneous formation of a functional barrier from tight junction proteins also associated with the purse string.
Figures

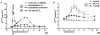
References
-
- Albers TM, Lomakina I, Moore RP. Structural and functional roles of cytoskeletal proteins during repair of native guinea pig intestinal epithelium. Cell Biology International. 1996;20:821–830. - PubMed
-
- Bement WM, Mandato CA, Kirsch MN. Wound-induced assembly and closure of an actomyosin purse string in Xenopus oocytes. Current Biology. 1999;9:579–587. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

