STAT1 deficiency unexpectedly and markedly exacerbates the pathophysiological actions of IFN-alpha in the central nervous system
- PMID: 12461178
- PMCID: PMC138590
- DOI: 10.1073/pnas.252454799
STAT1 deficiency unexpectedly and markedly exacerbates the pathophysiological actions of IFN-alpha in the central nervous system
Abstract
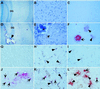
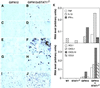
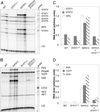
Although signal transducer and activator of transcription 1 (STAT1) is an essential signaling molecule in many IFN-alpha-regulated processes, some biological responses to IFN-alpha can occur independently of STAT1. To establish the role of STAT1 in mediating the biological actions of IFN-alpha in the CNS, transgenic mice [termed glial fibrillary acidic protein (GFAP)-IFN-alpha] with astrocyte production of IFN-alpha were bred to be null for the STAT1 gene. Surprisingly, GFAP-IFN-alpha mice deficient for STAT1 developed earlier onset and more severe neurological disease with increased lethality compared with GFAP-IFN-alpha mice sufficient for STAT1. Whereas the brain of 2- to 3-month-old GFAP-IFN-alpha mice showed little, if any abnormality, the brain from GFAP-IFN-alpha mice deficient for STAT1 had severe neurodegeneration, inflammation, calcification with increased apoptosis, and glial activation. However, the cerebral expression of a number of IFN-regulated STAT1-dependent genes increased in GFAP-IFN-alpha mice but was reduced markedly in GFAP-IFN-alpha STAT1-null mice. Of many other signaling molecules examined, STAT3 alone was activated significantly in the brain of GFAP-IFN-alpha STAT1-null mice. Thus, in the absence of STAT1, alternative signaling pathways mediate pathophysiological actions of IFN-alpha in the living brain, giving rise to severe encephalopathy. Finally, STAT1 or a downstream component of the JAKSTAT pathway may protect against such IFN-alpha-mediated injury in the CNS.
Figures

Similar articles
-
Interferon-independent, human immunodeficiency virus type 1 gp120-mediated induction of CXCL10/IP-10 gene expression by astrocytes in vivo and in vitro.J Virol. 2001 Aug;75(15):7067-77. doi: 10.1128/JVI.75.15.7067-7077.2001. J Virol. 2001. PMID: 11435587 Free PMC article.
-
Type I interferon-regulated gene expression and signaling in murine mixed glial cells lacking signal transducers and activators of transcription 1 or 2 or interferon regulatory factor 9.J Biol Chem. 2017 Apr 7;292(14):5845-5859. doi: 10.1074/jbc.M116.756510. Epub 2017 Feb 17. J Biol Chem. 2017. PMID: 28213522 Free PMC article.
-
Dysregulated Sonic hedgehog signaling and medulloblastoma consequent to IFN-alpha-stimulated STAT2-independent production of IFN-gamma in the brain.J Clin Invest. 2003 Aug;112(4):535-43. doi: 10.1172/JCI18637. J Clin Invest. 2003. PMID: 12925694 Free PMC article.
-
Innate STAT1-dependent genomic response of neurons to the antiviral cytokine alpha interferon.J Virol. 2005 Jul;79(13):8295-302. doi: 10.1128/JVI.79.13.8295-8302.2005. J Virol. 2005. PMID: 15956575 Free PMC article.
-
Interferon-gamma inhibits interferon-alpha signalling in hepatic cells: evidence for the involvement of STAT1 induction and hyperexpression of STAT1 in chronic hepatitis C.Biochem J. 2004 Apr 1;379(Pt 1):199-208. doi: 10.1042/BJ20031495. Biochem J. 2004. PMID: 14690454 Free PMC article.
Cited by
-
Gene-Expression Profiling Suggests Impaired Signaling via the Interferon Pathway in Cstb-/- Microglia.PLoS One. 2016 Jun 29;11(6):e0158195. doi: 10.1371/journal.pone.0158195. eCollection 2016. PLoS One. 2016. PMID: 27355630 Free PMC article.
-
Systemic interferon-alpha regulates interferon-stimulated genes in the central nervous system.Mol Psychiatry. 2008 Mar;13(3):293-301. doi: 10.1038/sj.mp.4002013. Epub 2007 May 8. Mol Psychiatry. 2008. PMID: 17486106
-
Protein arginine methyltransferases as regulators of cellular stress.Exp Neurol. 2025 Feb;384:115060. doi: 10.1016/j.expneurol.2024.115060. Epub 2024 Nov 17. Exp Neurol. 2025. PMID: 39551462 Free PMC article. Review.
-
IFN-gamma promotes complement expression and attenuates amyloid plaque deposition in amyloid beta precursor protein transgenic mice.J Immunol. 2010 May 1;184(9):5333-43. doi: 10.4049/jimmunol.0903382. Epub 2010 Apr 5. J Immunol. 2010. PMID: 20368278 Free PMC article.
-
The type I interferon-alpha mediates a more severe neurological disease in the absence of the canonical signaling molecule interferon regulatory factor 9.J Neurosci. 2010 Jan 20;30(3):1149-57. doi: 10.1523/JNEUROSCI.3711-09.2010. J Neurosci. 2010. PMID: 20089923 Free PMC article.
References
-
- Pestka S., Langer, J. A., Zoon, K. C. & Samuel, C. E. (1987) Annu. Rev. Biochem. 56, 727-777. - PubMed
-
- Belardelli F. (1995) Acta Pathol. Microbiol. Immunol. Scand. 103, 161-179. - PubMed
-
- Rho M. B., Wesselingh, S., Glass, J. D., McArthur, J. C., Choi, S., Griffin, J. & Tyor, W. R. (1995) Brain Behav. Immunol. 9, 366-377. - PubMed
-
- Lebon P., Badoual, J., Ponsot, G., Goutieres, F., Hemeury-Cukier, F. & Aicardi, J. (1988) J. Neurol. Sci. 84, 201-208. - PubMed
-
- Huang X., Yuang, J., Goddard, A., Foulis, A., James, R. F., Lernmark, A., Pujol-Borrell, R., Rabinovitch, A., Somoza, N. & Stewart, T. A. (1995) Diabetes 44, 658-664. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

