T cell activation causes diarrhea by increasing intestinal permeability and inhibiting epithelial Na+/K+-ATPase
- PMID: 12464679
- PMCID: PMC151630
- DOI: 10.1172/JCI15695
T cell activation causes diarrhea by increasing intestinal permeability and inhibiting epithelial Na+/K+-ATPase
Abstract
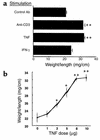
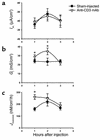
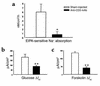
Inflammatory bowel disease (IBD) is associated with mucosal T cell activation and diarrhea. We found that T cell activation with anti-CD3 mAb induces profound diarrhea in mice. Diarrhea was quantified by intestinal weight-to-length (wt/l) ratios, mucosal Na(+)/K(+)-ATPase activity was determined and ion transport changes were measured in Ussing chambers. Anti-CD3 mAb increased jejunal wt/l ratios by more than 50% at 3 hours, returning to base line after 6 hours. Fluid accumulation was significantly reduced in TNF receptor-1 (TNFR-1(-/-)), but not IFN-gamma knockout mice. Anti-CD3 mAb decreased mucosal Na(+)/K(+)-ATPase activity, which was blocked by anti-TNF mAb and occurred to a lesser degree in TNFR-1(-/-) mice. Neither alpha nor beta subunits of Na(+)/K(+)-ATPase decreased in abundance at 3 hours. Intestinal tissue from anti-CD3-treated mice exhibited increased permeability to mannitol at 1 hour and decreases in electroneutral Na(+) absorption, Na(+)-dependent glucose absorption, and cAMP-stimulated anion secretion at 3 hours. Furthermore, enteral fluid accumulation was observed in CFTR(-/-) mice, indicating a minor role of active anion secretion. These data suggest that diarrhea in IBD is due to TNF-mediated malabsorption rather than to secretory processes. T cell activation induces luminal fluid accumulation by increasing mucosal permeability and reducing epithelial Na(+)/K(+)-ATPase activity leading to decreased intestinal Na(+) and water absorption.
Figures
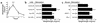

References
-
- Ciancio MJ, Chang EB. Epithelial secretory response to inflammation. Ann NY Acad Sci. 1992;664:210–221. - PubMed
-
- Baert FJ, et al. Tumor necrosis factor-alpha antibody (Infliximab) therapy profoundly down-regulates the inflammation in Crohn’s ileocolitis. Gastroenterology. 1999;116:22–28. - PubMed
-
- Bell SJ, Kamm MA. Antibodies to tumor necrosis factor alpha as treatment for Crohn’s disease. Lancet. 2000;355:858–860. - PubMed
-
- van Dullemen HM, et al. Treatment of Crohn’s disease with anti-tumor necrosis factor chimeric monoclonal antibody (cA2) Gastroenterology. 1995;109:129–135. - PubMed
-
- Casellas F, et al. Intraluminal colonic release of immunoreactive tumour necrosis factor in chronic ulcerative colitis. Clin Sci (Lond) 1994;87:453–458. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

