Active and passive membrane properties of rat sympathetic preganglionic neurones innervating the adrenal medulla
- PMID: 12482898
- PMCID: PMC2290734
- DOI: 10.1113/jphysiol.2002.023390
Active and passive membrane properties of rat sympathetic preganglionic neurones innervating the adrenal medulla
Abstract
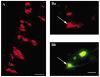
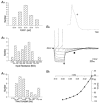
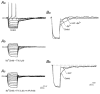
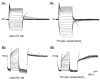
The intravascular release of adrenal catecholamines is a fundamental homeostatic process mediated via thoracolumbar spinal sympathetic preganglionic neurones (AD-SPN). To understand mechanisms regulating their excitability, whole-cell patch-clamp recordings were obtained from 54 retrogradely labelled neonatal rat AD-SPN. Passive membrane properties included a mean resting membrane potential, input resistance and time constant of -62 +/- 6 mV, 410 +/- 241 MOmega and 104 +/- 53 ms, respectively. AD-SPN were homogeneous with respect to their active membrane properties. These active conductances included transient outward rectification, observed as a delayed return to rest at the offset of the membrane response to hyperpolarising current pulses, with two components: a fast 4-AP-sensitive component (A-type conductance), contributing to the after-hyperpolarisation (AHP) and spike repolarisation; a slower prolonged Ba(2+)-sensitive component (D-like conductance). All AD-SPN expressed a Ba(2+)-sensitive instantaneous inwardly rectifying conductance activated at membrane potentials more negative than around -80 mV. A potassium-mediated, voltage-dependent sustained outward rectification activated at membrane potentials between -35 and -15 mV featured an atypical pharmacology with a component blocked by quinine, reduced by low extracellular pH and arachidonic acid, but lacking sensitivity to Ba(2+), TEA and intracellular Cs(+). This quinine-sensitive outward rectification contributes to spike repolarisation. Following block of potassium conductances by Cs(+) loading, AD-SPN revealed the capability for autorhythmicity and burst firing, mediated by a T-type Ca(2+) conductance. These data suggest the output capability is dynamic and diverse, and that the range of intrinsic membrane conductances expressed endow AD-SPN with the ability to generate differential and complex patterns of activity. The diversity of intrinsic membrane properties expressed by AD-SPN may be key determinants of neurotransmitter release from SPN innervating the adrenal medulla. However, factors other than active membrane conductances of AD-SPN must ultimately regulate the differential ratio of noradrenaline (NA) versus adrenaline (A) release secreted in response to various physiological and environmental demands.
Figures







Similar articles
-
Electrophysiological, pharmacological and molecular profile of the transient outward rectifying conductance in rat sympathetic preganglionic neurons in vitro.Neuroscience. 2011 Mar 31;178:68-81. doi: 10.1016/j.neuroscience.2010.12.061. Epub 2011 Jan 4. Neuroscience. 2011. PMID: 21211550
-
Membrane properties and synaptic potentials in rat sympathetic preganglionic neurons studied in horizontal spinal cord slices in vitro.J Auton Nerv Syst. 1995 May 17;53(1):1-15. doi: 10.1016/0165-1838(94)00161-c. J Auton Nerv Syst. 1995. PMID: 7673598
-
Afterdepolarization mechanism in the in vitro, cesium-loaded, sympathetic preganglionic neuron of the cat.J Neurophysiol. 1987 May;57(5):1325-37. doi: 10.1152/jn.1987.57.5.1325. J Neurophysiol. 1987. PMID: 3585470
-
Bulbospinal catecholamine neurones and sympathetic pattern generation.J Physiol Pharmacol. 1995 Sep;46(3):259-71. J Physiol Pharmacol. 1995. PMID: 8527808 Review.
-
Roles of Na+, Ca2+, and K+ channels in the generation of repetitive firing and rhythmic bursting in adrenal chromaffin cells.Pflugers Arch. 2018 Jan;470(1):39-52. doi: 10.1007/s00424-017-2048-1. Epub 2017 Aug 3. Pflugers Arch. 2018. PMID: 28776261 Free PMC article. Review.
Cited by
-
Plateau potentials and membrane oscillations in parasympathetic preganglionic neurones and intermediolateral neurones in the rat lumbosacral spinal cord.J Physiol. 2005 Mar 1;563(Pt 2):583-96. doi: 10.1113/jphysiol.2004.076802. Epub 2004 Dec 23. J Physiol. 2005. PMID: 15618277 Free PMC article.
-
Heterogeneity of membrane properties in sympathetic preganglionic neurons of neonatal mice: evidence of four subpopulations in the intermediolateral nucleus.J Neurophysiol. 2010 Jan;103(1):490-8. doi: 10.1152/jn.00622.2009. Epub 2009 Nov 18. J Neurophysiol. 2010. PMID: 19923248 Free PMC article.
-
The effect of hyperpolarization-activated cyclic nucleotide-gated ion channel inhibitors on the vagal control of guinea pig airway smooth muscle tone.Br J Pharmacol. 2014 Aug;171(15):3633-50. doi: 10.1111/bph.12745. Br J Pharmacol. 2014. PMID: 24762027 Free PMC article.
-
Maternal diabetes increases small conductance Ca2+-activated K+ (SK) currents that alter action potential properties and excitability of cardiac motoneurons in the nucleus ambiguus.J Neurophysiol. 2010 Oct;104(4):2125-38. doi: 10.1152/jn.00671.2009. Epub 2010 Jul 28. J Neurophysiol. 2010. PMID: 20668269 Free PMC article.
-
Maternal diabetes increases large conductance Ca2+-activated K+ outward currents that alter action potential properties but do not contribute to attenuated excitability of parasympathetic cardiac motoneurons in the nucleus ambiguus of neonatal mice.Am J Physiol Regul Integr Comp Physiol. 2011 May;300(5):R1070-8. doi: 10.1152/ajpregu.00470.2010. Epub 2011 Jan 19. Am J Physiol Regul Integr Comp Physiol. 2011. PMID: 21248308 Free PMC article.
References
-
- An WF, Bowlby MR, Betty M, Cao J, Ling HP, Mendoza G, Hinson JW, Mattsson KI, Strassle BW, Trimmer JS, Rhodes KJ. Modulation of A-type potassium channels by a family of calcium sensors. Nature. 2000;403:553–556. - PubMed
-
- Backman SB, Sequeira-Martinho H, Henry JL. Adrenal versus nonadrenal sympathetic preganglionic neurones in the lower thoracic intermediolateral nucleus of the cat: physiological properties. Canadian Journal of Physiology and Pharmacology. 1990;68:1447–1456. - PubMed
-
- Bahring R, Dannenberg J, Peters HC, Leicher T, Pongs O, Isbrandt D. Conserved Kv4 N-terminal domain critical for effects of Kv channel-interacting protein 2. 2 on channel expression and gating. Journal of Biological Chemistry. 2001;276:23 888–23 894. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

